This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Gene variations for immune and metabolic conditions have persisted in humans for more than 700,000 years
Like a merchant of old, balancing the weights of two different commodities on a scale, nature can keep different genetic traits in balance as a species evolves over millions of years.
These traits can be beneficial (for example, fending off disease) or harmful (making humans more susceptible to illness), depending on the environment.
The theory behind these evolutionary trade-offs is called balancing selection. A University at Buffalo-led study published in eLife explores this phenomenon by analyzing thousands of modern human genomes alongside ancient hominin groups, such as Neanderthal and Denisovan genomes.
The research has "implications for understanding human diversity, the origin of diseases, and biological trade-offs that may have shaped our evolution," says evolutionary biologist Omer Gokcumen, the study's corresponding author.
Gokcumen, Ph.D., associate professor of biological sciences in the UB College of Arts and Sciences, adds that the study shows that many biologically relevant variants "have been segregating among our ancestors for hundreds of thousands, or even millions of years. These ancient variations are our shared legacy as a species."
Ties with Neanderthals stronger than previously thought
The work builds upon genetic discoveries in the past decade, including when scientists uncovered that modern humans and Neanderthals interbred as early humans moved out of Africa.
It also coincides with the growth of personalized genetic testing, with many people now claiming that a small percentage of their genome comes from Neanderthals. But, as the eLife study shows, humans share much more in common with Neanderthals than those small percentages indicate.
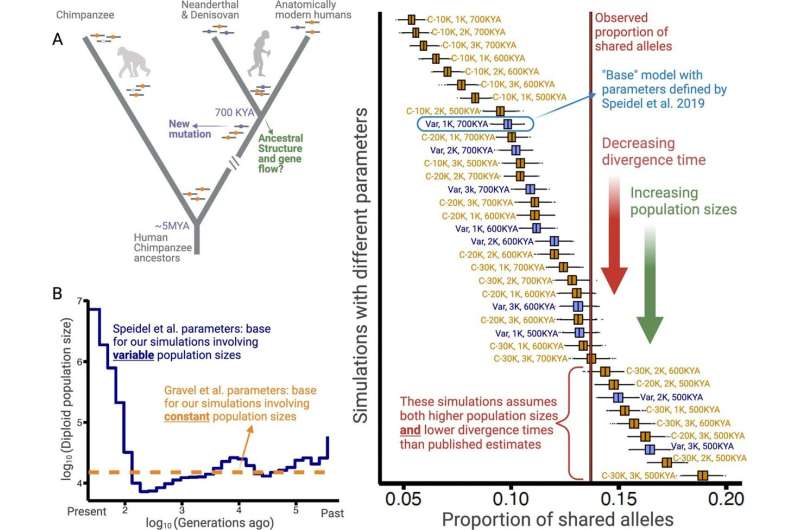
This additional sharing can be traced back to a common ancestor of Neanderthals and humans that lived about 700,000 years ago. This common ancestor bequeathed to the Neanderthals and modern humans a shared legacy in the form of genetic variation.
The research team explored this ancient genetic legacy, focusing on a particular type of genetic variation: deletions.
Gokcumen says that the "deletions are strange because they affect large segments. Some of us are missing large chunks of our genome. These deletions should have negative effects, and as a result, be eliminated from the population by natural selection. However, we observed that some deletions are older than modern humans, dating back millions of years ago."
Gene variations passed down over millions of years
The researchers used computational models to show an excess of these ancient deletions, some of which have persisted since our ancestors first learned to make tools, some 2.6 million years ago. Furthermore, the models found that balancing selection can explain this surplus of ancient deletions.
"Our study contributes to the growing body of evidence suggesting that balancing selection may be an important force in the evolution of genomic variation among humans," says first author Alber Aqil, a Ph.D. candidate in biological sciences in Gokcumen's lab.
The investigators found that deletions dating back millions of years are more likely to play an outsized role in metabolic and autoimmune conditions.
Indeed, the persistence of versions of genes that cause severe disease in human populations has long baffled scientists since they expect natural selection to get rid of these versions of genes. It is, after all, very unusual for potentially disease-causing variation to persist for such long periods. The authors argue that balancing selection can solve this riddle.
Aqil says that these variations may "protect against infectious diseases, outbreaks, and starvation, which have occurred periodically throughout human history. Thus, the findings represent a considerable leap in our understanding of how genetic variations evolve in humans. A variant may be protective against a pathogen or starvation while also underlying certain metabolic or autoimmune disorders, like Crohn's disease."
More information: Alber Aqil et al, Balancing selection on genomic deletion polymorphisms in humans, eLife (2023). DOI: 10.7554/eLife.79111
Journal information: eLife
Provided by University at Buffalo