New technique to make complex molecules for antibiotics and anti-fungal medicines
Scientists invented a pioneering technique to construct molecules discovered in rare sediments from the Bahamas with the potential to help treat disease and infection
Scientists have created a much faster way to make certain complex molecules, which are widely used by pharmaceuticals for antibiotics and anti-fungal medicines.
The first-of-its-kind discovery by chemists at the University of Bristol has the potential to speed up the production of such drugs, making them cheaper and more accessible.
The breakthrough, published in Nature Chemistry, marks the culmination of a five-year research project which has finally cracked how to reconstruct in a laboratory a particularly complex molecule, from the family of molecules known as the polyketides.
Lead author Sheenagh Aiken, a Ph.D. student at the university's School of Chemistry when the work was completed, said, "It's an exciting discovery, which could bring important benefits for the pharmaceutical industry and public health.
"We chose this specific polyketide because it is one of the hardest to work with and manipulate. Now we've devised a way to make it more quickly in the lab, this should make it easier to apply the technique to others with equally significant implications."
Polyketides are natural products found in a variety of places, including bacteria, sea sponges and sediments. The compounds are often only present in tiny quantities, but they have powerful medicinal properties which the pharmaceutical industry has used to develop a wide range of medicines. Around a fifth of all pharmaceuticals are derived from polyketides.
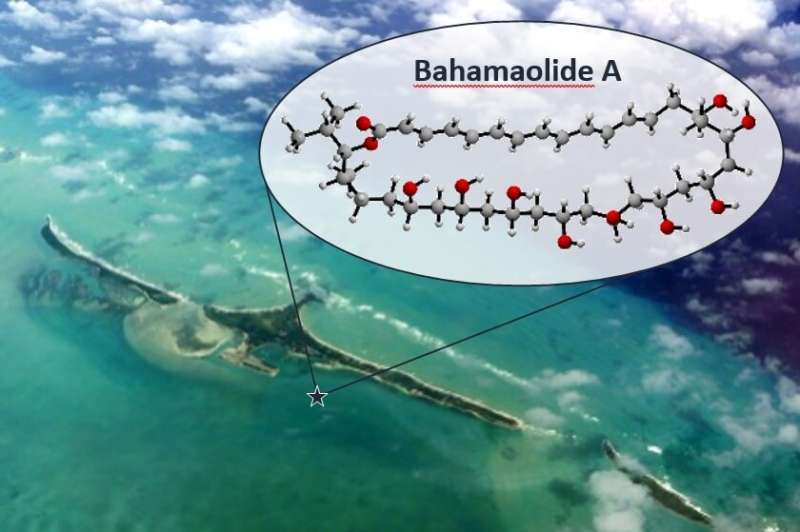
The research team worked on a polyketide called Bahamaolide A, which acquired its name as it originates from bacteria cultured from a marine sediment at North Cat Cay in the Bahamas.
Using existing methods, it would normally take more than 20 different steps to construct it in a laboratory. The researchers found a new, improved way to combine the building blocks for the molecule so it could be made in just 14 steps.
Sheenagh said, "The technique mimics nature by coupling together building blocks and using catalysts to add and modify functional groups in a process like an assembly line. This highly controlled and predictable approach has potential for making the manufacture of sophisticated molecules more efficient."
Professor Varinder Aggarwal, from the University of Bristol, who directed the research added, "Due to their highly specific and potent biological activity as well their structural complexity, polyketide natural products have been attractive targets in chemical synthesis research for over 50 years. Through a different strategy to what others have done before, we have succeeded in developing a step change in efficiency to this important class of molecules."
More information: Sheenagh G. Aiken et al, Iterative synthesis of 1,3-polyboronic esters with high stereocontrol and application to the synthesis of bahamaolide A, Nature Chemistry (2022). DOI: 10.1038/s41557-022-01087-9
Journal information: Nature Chemistry
Provided by University of Bristol