A sustainable path for energy-demanding photochemistry
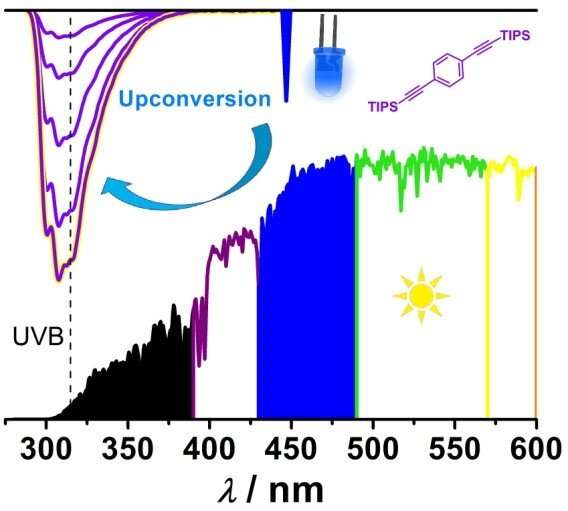
Many photochemical processes rely on UV light from inefficient or toxic light sources that the LED technology cannot replace for technical reasons. An international team of scientists led by Professor Christoph Kerzig of Johannes Gutenberg University Mainz (JGU) in Germany and Professor Nobuhiro Yanai of Kyushu University in Japan has now developed the first molecular system for the conversion of blue light into high-energy UV photons with wavelengths below 315 nanometers.
These photons in the so-called UVB range are essential for numerous photochemical processes in the context of light-to-energy conversion, disinfection, or even wastewater treatment applications. However, sunlight cannot provide UVB photons, and their artificial generation typically relies on mercury lamps or other highly inefficient alternatives.
The new findings show that a metal-free photon upconversion (UC) system can transform readily available visible light into UVB photons. Hence, this breakthrough can be regarded as a more environmentally friendly approach. Initial mercury-free applications have already been demonstrated in the lab.
Collaborative research with a long tradition
Both research groups started working on upconversion several years ago. UC is a process in which the absorption of two photons of lower energy leads to the emission of one photon of higher energy. This technique has been developed to increase the efficiency of solar cells, mainly by converting low-energy photons in the infrared region.
"In contrast, highly energetic UV photons are within reach when blue light is used as the energy source," explained Professor Kerzig of the Department of Chemistry at Mainz University.
Tailor-made molecules have been prepared in Mainz and characterized with a new large-scale laser device recently installed in the Kerzig group. Furthermore, special spectroscopic techniques in the lab of Professor Nobuhiro Yanai have been applied to the UC system to understand its performance in detail.
While the current paper represents the first collaboration between the Kerzig and Yanai groups, the chemistry departments of both universities have a well-established student exchange program. This novel collaboration will further strengthen the network between Mainz and Kyushu.
Development of reusable upconversion materials
The scientists used a commercial blue LED as light source and exploited the generated UV light for the cleavage of strong chemical bonds that would otherwise require very harsh reaction conditions. Moreover, using the laser setup in Mainz, Ph.D. student Till Zähringer managed to observe all intermediates in the complex energy conversion mechanism.
"Our next goal is to develop reusable materials for versatile applications," said Professor Nobuhiro Yanai.
His group in Kyushu is well known for the development of photoactive materials. The combination of materials science, photochemistry, and photocatalysis in the framework of the Kyushu-Mainz collaboration will pave the way for this ambitious goal.
The research is published in the journal Angewandte Chemie International Edition.
More information: Till J. B. Zähringer et al, Blue‐to‐UVB Upconversion, Solvent Sensitization and Challenging Bond Activation Enabled by a Benzene‐Based Annihilator, Angewandte Chemie International Edition (2022). DOI: 10.1002/anie.202215340
Journal information: Angewandte Chemie International Edition
Provided by Universitaet Mainz