Research team introduces new technology for analysis of protein activity in cells

Proteins constitute principal building blocks in all living organisms. They are often described as the workers of the cell, where they—together or individually—perform numerous essential tasks. If something goes wrong, the consequences are often serious. Both research and health care have expressed the need for effective tools to analyze the functions and activities of proteins, and in a new article in scientific journal Nature Communications, Professor Ola Söderberg's team introduces MolBoolean, a technology that is expected to open important doors in cell and cancer research.
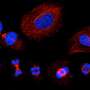
"MolBoolean is a method to determine levels of a pair of proteins in individual cells, while in parallel identifying the percentage of these proteins that bind each other. We have continued to develop a tool that we created in 2006, which today is used all over the world, and the functions that we are now adding generate information about the relative amounts of different proteins as well as relative amounts of protein complexes, thus making the cells' activity status and communication more visible," says Ola Söderberg, Professor of Pharmaceutical Cell Biology at Uppsala University. "This can be compared to assessing a restaurant; 10 positive reviews give some information, but it is important to know whether they are based on ten or 1,000 opinions,"
The group's results are piquing great interest in both academia and industry, and currently the Stockholm-based company Atlas Antibodies—which recently overtook the patent for MolBoolean—is preparing the method for the market. The tool is expected to be of great value in research, and in the long term, also in health care to improve diagnosis and choice of treatment for cancer.
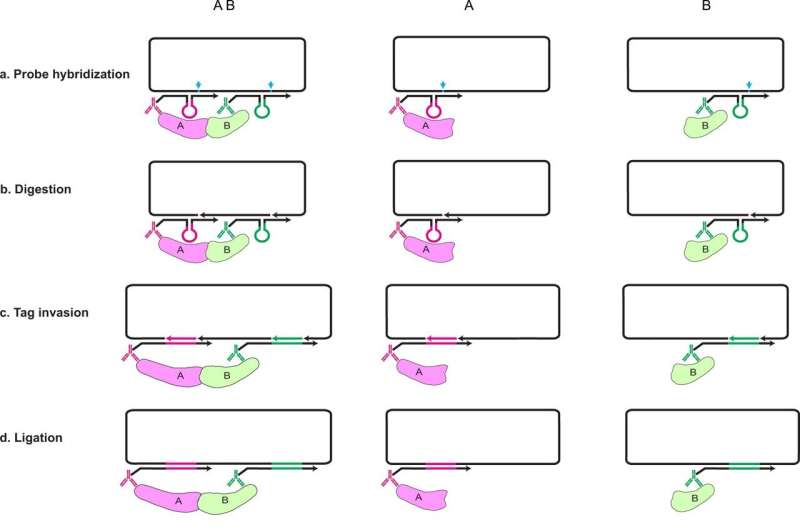
"Our research aims to develop tools to visualize processes within cells and to increase, for example, the knowledge of what happens in cancer cells. Knowing the balance between free and interacting proteins is important when studying cell signaling, and we believe that MolBoolean will be appreciated in many research laboratories," states Doroteya Raykova, researcher at the Department of Pharmaceutical Biosciences and first author of the article.
MolBoolean is developed by Ola Söderberg's group at Uppsala University's Faculty of Pharmacy. The work has been carried out in collaboration with researchers at the universities of Porto and Uppsala, SciLifeLab and Atlas Antibodies.
More information: Doroteya Raykova et al, A method for Boolean analysis of protein interactions at a molecular level, Nature Communications (2022). DOI: 10.1038/s41467-022-32395-w
Journal information: Nature Communications
Provided by Uppsala University