Catching up with quicksilver: MXene material can counter mercury contamination

Researchers estimate that mercury emissions in the atmosphere have quadrupled since the Industrial Revolution. The heavy metal, generated by burning fossil fuels and the disposal of industrial and medical waste, has become so persistent in aquatic environments that the U.S. Food and Drug Administration suggests about a half dozen species of fish are so mercury-contaminated that people should avoid consuming them. Researchers have been working for many years to develop systems for removing mercury from water. But a team at Drexel University might have found just the right material to efficiently catch the evasive quicksilver—even at low levels—and clean up contaminated bodies of water.
Among many methods of removing mercury from water, adsorption—the process of chemically attracting and removing contaminants—is the most promising technology due to its relative simplicity, efficiency and low cost, according to Drexel College of Engineering Professor Masoud Soroush, Ph.D., whose lab is developing a new adsorption technology.
"Modern adsorbents, such as resins, mesoporous silica, chalcogenides, and mesoporous carbons, have higher efficiencies than traditional adsorbents, such as activated carbon, clays, and zeolites that have low affinity toward mercury and low capacities," Soroush said. "However, the problem with all these materials, is that their mercury-removal efficiencies are still low, and they are unable to lower mercury level to less than 1 part per billion."
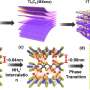
Soroush's team of researchers from Drexel and Temple University has explored synthesizing and using a surface-modified titanium carbide MXene for mercury removal. MXenes are a family of two-dimensional nanomaterials that was discovered at Drexel more than a decade ago and has demonstrated many exceptional properties. The team recently reported its results in the Journal of Hazardous Materials.
For mercury ion removal, titanium carbide MXene's advantages, according to Soroush, are its negatively charged surface and the tunability and versatility of its surface chemistry, rendering the MXene attractive for heavy metal ion removal. Because of these properties and the layered structure of the MXene, titanium carbide MXene-based materials have shown superior performance in gas separation, removing salt from water, killing bacteria, and kidney dialysis.
"We knew that 2D materials, such as graphene oxide and molybdenum disulfide, had previously been effective in removing heavy metals from wastewater through adsorption because of their chemical functionalities/structures that attract metal ions," Soroush said. "MXenes are a similar type of materials but we estimated that titanium carbide MXene could have much greater uptake capacities than these other materials—therefore making it a better sorbent for mercury ions."
But Soroush's team needed to make a key adjustment to titanium carbide MXene's chemical structure to further improve the material for one of its most challenging tasks.
"Mercury is called quicksilver for a reason—it's quite evasive once emitted into the environment, whether by burning fossil fuels, mining, or waste incineration," Soroush said. "It quickly changes its chemical form—increasing its toxicity and making it tremendously difficult to remove from the bodies of water where it inevitably accumulates. So, to attract mercury ions even faster we needed to modify the surface of titanium carbide MXene flakes."
There is a natural attraction between mercury ions and titanium carbide MXene surface, as metal ions, like mercury, are positively charged and the surface of the MXene flakes are negatively charged. However, to pull mercury ions out of water more strongly the team needed to give this attraction a boost. To this end, they treated the MXene flakes with chloroacetic acid—a process called carboxylation—which provides the MXene with highly mobile, strong carboxylic acid groups and increases the MXene-flakes surface negative charge, improving the ability of the flakes to attract and retain mercury ions.
The result was a new sorbent material—carboxylated titanium carbide MXene, that demonstrated a faster mercury-ion uptake and greater capacity than all commercially available adsorbents, according to the researchers.
"Carboxylated titanium carbide MXene proved to be far superior to sorbent material currently being used for mercury-ion removal," Soroush said. "Within one minute it was able to remove 95% of mercury ions from a water sample contaminated at a concentration of 50 parts per million, which means it could be effective and efficient enough for use in large scale wastewater treatment".
Within five minutes, titanium carbide MXene and carboxylated titanium carbide MXene removed 98% of mercury ions from a 10-milliliter water sample contaminated with mercury ions at concentrations between 1 and 1000 parts per million.
"This indicates that both [MXene] and [carboxylated MXene] are effective adsorbents to remove mercury ions from wastewater due to their special structural properties and high density of surface functional groups," the team wrote. "Generally, the adsorption mechanism of metal ions follows two steps; at first, the ions are quickly adsorbed on the available active sites, and the process is swift. The adsorption proceeds slower as the adsorption sites fill up, and the ions are required to diffuse into the pores and interlayer."
The development is significant in the battle to contain mercury pollution, which has become so pervasive that health authorities recommend avoiding eating certain species of fish altogether. Efforts to contain the mercury released by burning fossil fuels has proven as challenging as reducing dependence on the fuels themselves.
While shifting away from the polluting energy sources is the ultimate solution to preventing the release of heavy metals, like mercury into the environment, Soroush suggests that this breakthrough could lead new possibilities for cleaning up the pollution that has already been created.
"We envision that the use of the carboxylated MXene technology to remove all heavy metal ions," he said. "Besides using the carboxylated MXene as a sorbent, another way of achieving this is to fabricate filters coated or embedded with the carboxylated MXene."
More information: Ali Pournaghshband Isfahani et al, Efficient mercury removal from aqueous solutions using carboxylated Ti3C2T MXene, Journal of Hazardous Materials (2022). DOI: 10.1016/j.jhazmat.2022.128780
Li Ding et al, MXene molecular sieving membranes for highly efficient gas separation, Nature Communications (2018). DOI: 10.1038/s41467-017-02529-6
Kazem Meidani et al, Titanium Carbide MXene for Water Desalination: A Molecular Dynamics Study, ACS Applied Nano Materials (2021). DOI: 10.1021/acsanm.1c00944
One-Sun Lee et al, Antibacterial Mechanism of Multifunctional MXene Nanosheets: Domain Formation and Phase Transition in Lipid Bilayer, Nano Letters (2021). DOI: 10.1021/acs.nanolett.1c01986
Fayan Meng et al, MXene Sorbents for Removal of Urea from Dialysate: A Step toward the Wearable Artificial Kidney, ACS Nano (2018). DOI: 10.1021/acsnano.8b06494
Journal information: Nature Communications , Nano Letters , Journal of Hazardous Materials , ACS Nano
Provided by Drexel University