Achieving higher performance with potassium ion battery
Supercapacitors are emerging as alternatives to lithium-ion batteries, offering higher power densities and longer lifetimes (number of cycles where capacity is maintained). A supercapacitor is like a cross between a battery (with high energy storage) and a regular capacitor (with high power discharge).
New research from the City University of Hong Kong published March 21 in Nano Research Energy demonstrates outstanding performance of a capacitor built with MXene compounds. MXenes are two-dimensional inorganic compounds whose large molecular surface areas for energy storage give them ultrahigh conductivity and storage capacity.
Supercapacitors can store lots of energy in a small space and release it at a high current; for example, they can supply power for mini devices such as wearable electronics. However, when made with organic molecules, supercapacitors risk catching fire.
The new study explored supercapacitors made with inorganic MXene molecules to reduce fire risk. Instead of the more expensive lithium, they used potassium. The potassium ion or K-ion is one of the most frequently used electrolytes to allow electrical current to flow in a battery. Guojin Liang, lead author of the paper and researcher from the Department of Materials Science and Engineering says that they "have investigated the aqueous supercapacitors by utilizing the intrinsic safe water-based electrolytes and focused on K-ion storage, which is cheaper and more abundant in earth to benefit for safe and low-cost applications."
MXenes compounds consist of several-atoms-thick layers of transition metals, such as metal carbides, nitrides, or carbonitrides. They have the electrical properties of efficient electron transport across the conductive metal carbide layer, as well as a metal surface great for redox (electron transfer) reactions.
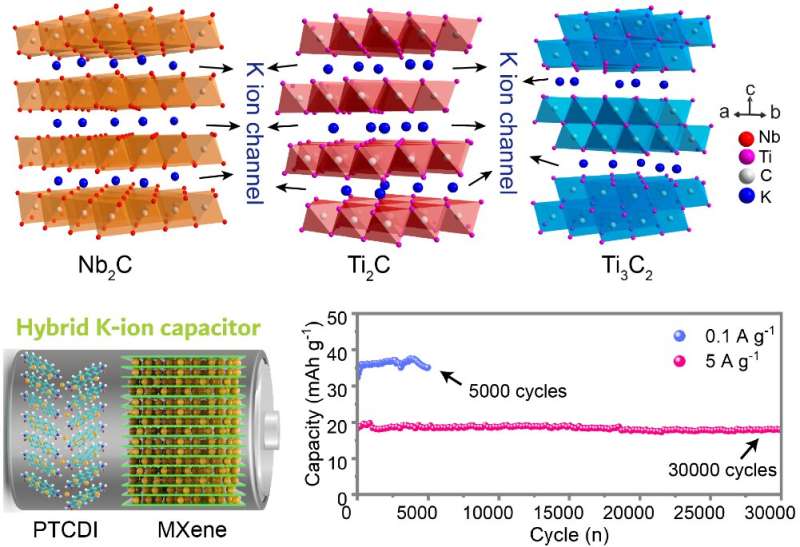
From the diverse MXenes, this study selected three for comparison of performance. "By horizontally comparing the K-ion storage performance of three representative MXene species, we want to figure out the relationship between the structure and their K-ion storage performance," says lead author Xinliang Li, also from the Department of Materials Science and Engineering.
The three MXene electrodes or electrical conductors—Nb2C, Ti2C and Ti3C2—were investigated for their electrochemical behaviors, including the chemistry of how the K-ions got inserted into the MXene layers as well as how ions adhered to the metal surfaces. The researchers evaluated the supercapacitors respect to storage mechanism, capacity, rate performance, and cyclic performance.
The K-ion capacitor with the Nb2C MXene had the most outstanding performance, with the highest power density (amount discharged) of 2336 W/kg and an energy density (amount stored) of 24.6 Wh/kg. While lithium-ion batteries have higher energy densities than capacitors, their power density is only in the 250-340 W/kg. A K-ion capacitor with MXene can, therefore, discharge power orders of magnitude faster. The capacitor with Nb2C MXene maintained nearly full capacity (94.6%) after 30,000 cycles of discharging 5 amperes/g of electricity, in contrast to the roughly 500 cycles a lithium-ion battery is expected to last.
All the MXene materials exhibited supercapacitor behaviors—fast kinetics and durable K-ion storage—delivering better performance than other K-ion host materials. The results stem from the stable structure of MXene as it gets and gives up potassium ions. Says Liang, "It could be ascribed to the intrinsically large interlayer distance for K-ion transport and the superb structural stability of MXene, even subjected to long-term potassiation/depotassiation process."
Even though only three MXene electrodes were investigated, other MXene compounds may have great potential to serve as aqueous K-ion host electrodes. The researchers hope their findings will "draw further attention to other promising MXene electrodes for durable K-ion storage."
The researchers plan to further experiment with MXene electrodes towards improved performance for practical applications. "Regarding the K-ion capacitor, we would like to modify and manipulate the MXene electrode species for higher energy density," says professor Chunyi Zhi. They ultimately aspire to refine K-ion capacitors for wearable electronics and other mini power devices, since they are high performing, safe, and relatively cheap.
More information: Yi-Chun Lu et al, Design strategies for low temperature aqueous electrolytes, Nano Research Energy (2022). DOI: 10.26599/NRE.2022.9120003
Provided by Tsinghua University Press