Dual temperature control system to regulate isoprene biosynthesis in baker's yeast
Baker's yeast, Saccharomyces cerevisiae, has been extensively engineered to produce a vast variety of chemicals that are not naturally produced by yeast, including the important monomer for synthetic rubber, isoprene. However, all organisms are naturally evolved for better survival rather than maximized biosynthesis of products of human interest. S. cerevisiae is not an exception. Impaired cell growth was observed during its engineering by pathway compartmentation for improved isoprene biosynthesis.
To address this issue, researchers from the College of Chemical and Biological Engineering of Zhejiang University have developed a temperature-responsive dynamic control system to regulate the initiation time of isoprene biosynthesis during the fermentation. This study was published online in Frontiers of Chemical Science and Engineering.
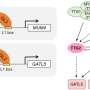
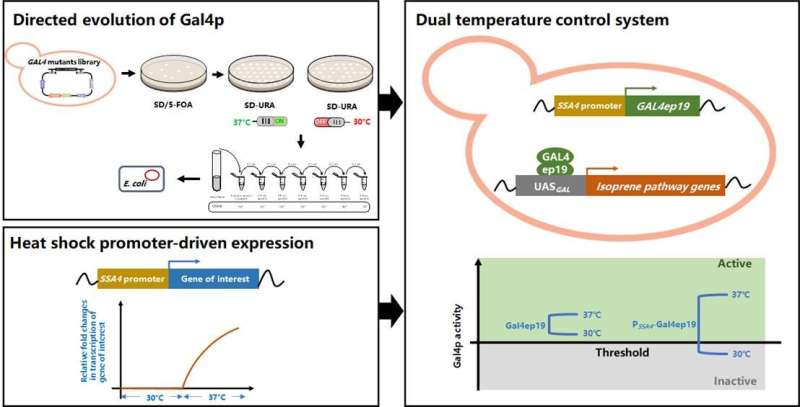
The native transcriptional activator Gal4p of S. cerevisiae was engineered to gain temperature sensitivity and meanwhile its expression was driven by a heat-shock promoter. In this way, a dual temperature regulation system was developed, the application of which led to limited expression of pathway genes at the optimal temperature for cell growth (30 °C) and enhanced gene expression when the culture temperature was switched to the optimal temperature for isoprene synthesis (37 °C).
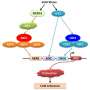
The "cold-sensitive" Gal4p mutant playing the key regulatory role was created by the Nobel prize-winning directed evolution technology. To facilitate fast and accurate selection of the mutants with temperature sensitivity from the random mutant library containing thousands of mutants, a growth-indicated high-throughput screening method was established based on the cytotoxicity of 5-fluorouridine formed by URA3-catalyzed conversion of 5-fluoro-orotic acid. The negative correlation between Gal4p activity at a certain temperature and the biomass of 5-fluorouridine-accumulating strains enabled selection of Gal4p mutants with lower activity at 30 °C and higher activity at 37 °C.
When the "cold-sensitive" Gal4p mutant was expressed under the control of a heat-shock promoter, its regulatory activity at the permissive temperature was further increased due to the enhanced expression level, and the basal expression of the pathway genes at the restrictive temperature was further reduced. Employment of this dual temperature control strategy led to 34.5% and 72% improvements in cell growth and isoprene production of S. cerevisiae, respectively. This study reports the creation of the first cold-sensitive variants of Gal4p by directed evolution and provides a dual temperature control system for yeast engineering that may also be conducive to the biosynthesis of other high-value natural products.
More information: Jiaxi Lin et al, Development of a dual temperature control system for isoprene biosynthesis in Saccharomyces cerevisiae, Frontiers of Chemical Science and Engineering (2021). DOI: 10.1007/s11705-021-2088-0
Provided by Higher Education Press