Large numbers of macrocyclic compounds needed for developing drugs against difficult disease targets synthesized
Scientists at EPFL have found a way to synthesize large numbers of macrocyclic compounds, which are needed for developing drugs against difficult disease targets.
When pharmaceutical companies begin looking for a drug candidate, they use a filtering process known as "high-throughput screening." Here, large numbers of different chemical compounds are tested to see which will bind to a protein that is the target of the disease they want to address.
Pharmaceutical companies actually have libraries of 1-2 million "small-molecule" compounds collected over years. But in many cases, screening classical small-molecule compounds can't identify drug candidates simply because they don't contain a compound that binds sufficiently strong to the target.
A solution has been found in the "macrocycles," an emerging class of molecules that have proven to be ideal for binding difficult targets like proteins with flat surfaces or even proteins bound to other proteins. The problem is that current macrocycle libraries only contain less than 10,000 compounds, which limits the chance of finding drug candidates that can bind a given disease target.
But a group of chemists at EPFL have now found a way to generate large numbers of macrocycles, which can significantly increase the sizes of available libraries. The breakthrough, published in Nature Communications, is the work of the group of Professor Christian Heinis at EPFL's School of Basic Sciences.
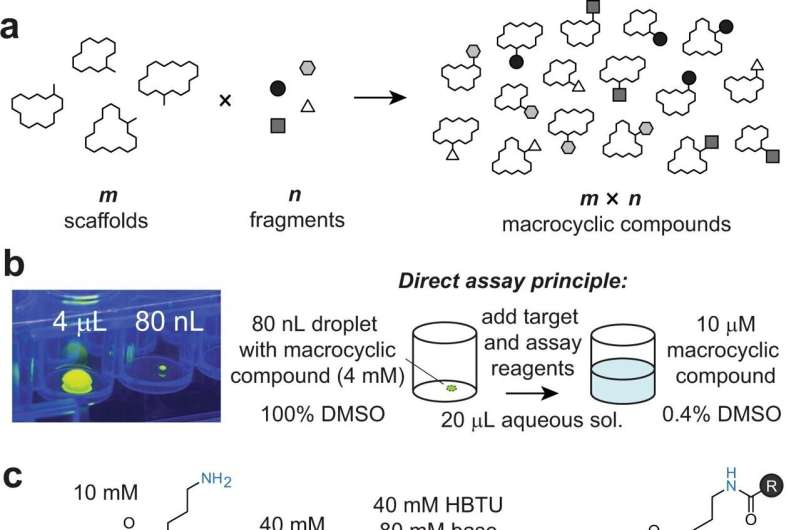
"Our approach is based on combining a large number 'm' of different macrocyclic scaffolds with a myriad of 'n' chemical fragments to generate 'm×n' different macrocyclic compounds," says Heinis. "For example, we generated a library of 19,968 macrocycles by reacting 192 macrocycle scaffolds with 104 carboxylic acid fragments."
With the help of EPFL's Biomolecular Screening Facility, the scientists performed the reactions in tiny volumes of 40 nanoliters and by transferring the reagents using acoustic waves, which is enormously fast. Through the miniaturization and the high speed, the library of 19,968 macrocyclic compounds was put together in just half a day.
To test the method, the Ph.D. student leading the project, Sevan Habeshian, used it to develop nanomolar inhibitors against thrombin and the MDM2:p53 protein-protein interaction, which are drug targets for strokes and cancer.
Working with scientists from the universities of Padova and Venice, the team got an X-ray structure of the thrombin inhibitor while it bound the protein. "The structural analysis validated the approach of screening compounds containing macrocyclic cores and laterally linked chemical fragments," says Habeshian.
"We are currently applying the approach for developing macrocyclic compounds to a range of disease targets for which pharmaceutical companies are struggling to generate drugs based on classical small molecules," says Heinis. "Given the small size and the limited polar surface of macrocyclic compounds, they have a high chance of being passing through cell membranes, which means that they can be used to developing drugs for intracellular targets or even drugs that are taken orally."
More information: Sevan Habeshian et al, Synthesis and direct assay of large macrocycle diversities by combinatorial late-stage modification at picomole scale, Nature Communications (2022). DOI: 10.1038/s41467-022-31428-8
Journal information: Nature Communications
Provided by Ecole Polytechnique Federale de Lausanne