Improving hydrogen peroxide production through sustainable photocatalysis
Hydrogen peroxide is used in many industries for a variety of purposes, including bleaching, sewage treatment, sterilization, and even as rocket fuel. Because hydrogen peroxide's byproduct is water, it has been lauded as a "green," environmentally friendly chemical, but a closer look at hydrogen peroxide's production process reveals a more complicated story. Problems like the amount of energy used for the production process and mining for the required chemicals have dramatic environmental impacts. As demand increases for hydrogen peroxide globally, researchers are trying to find new ways to produce hydrogen peroxide that are safer and better for the environment.
Previous research has identified techniques using photocatalysis, the use of light to start a chemical reaction, and hot electrons, which are high-energy electrons that have been charged through visible and infrared light, as alternative solutions to hydrogen peroxide production. Both photocatalysis and hot electrons have been used in green energy alternatives, such as solar power, in the past, but limitations on both processes have prevented them from being implemented for hydrogen peroxide production.
To address some of these limitations, researchers at Shanghai Jiao Tong University found ways to make the hot electrons last longer so that photocatalysis can be used to produce hydrogen peroxide in a safer, cleaner production process.
The findings were published on June 25 in Nano Research.
Paper author Xinhao Li, a professor in the School of Chemistry and Chemical Engineering at Shanghai Jiao Tong University, explained some of the limitations of using hot electrons in the production of hydrogen peroxide. "The lifetime of hot electrons, typically on a time scale of 0.4 to 0.3 picoseconds, could not be matched well with the time scale of typical chemical reactions, including the oxygen reduction reaction to hydrogen peroxide. Because of this, it is appealing to develop powerful methods to prolong the lifetime of thermalized hot carriers over cheap photocatalysts for hydrogen peroxide production using only water, air, and solar light," Li said.
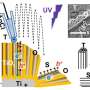
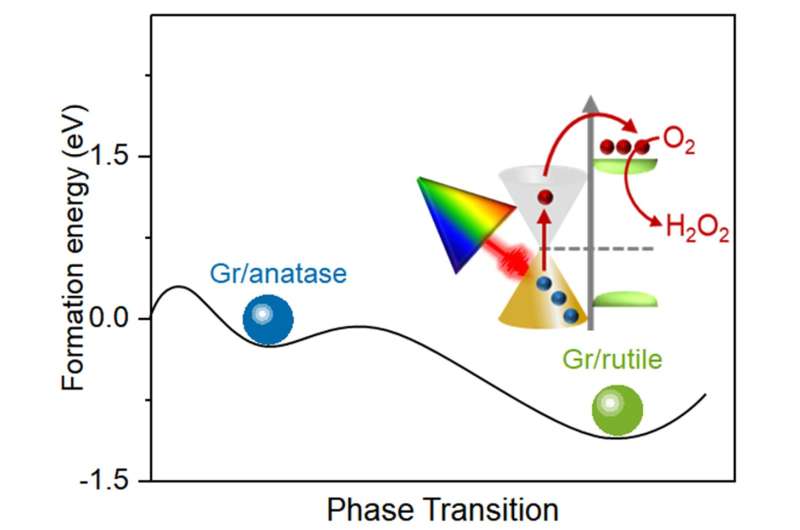
The method to maintain the energy of the hot electrons proposed by researchers is straightforward. A heterojunction—a combination of two different layers of semiconductors—of rutile titanium dioxide and graphene is made to harvest the hot electrons. First researchers explored ways to synthetically produce rutile titanium dioxide quickly and efficiently. It takes 24 hours for the phase transfer process by milling method to convert anatase titanium dioxide to rutile titanium dioxide, but researchers were able to reduce this to 5 minutes.
The combination of rutile titanium dioxide and graphene forms an elevated Schottky barrier, which is essential for prolonging the lifetime of hot electrons. A Schottky barrier forms between a metal and semiconductor and acts as a barrier for electrons. Because the Schottky barrier between rutile titanium dioxide and graphene is high, it facilitates the hot electron injection and prevents the electrons from flowing backwards through the barrier. The elevated barrier is achieved because of the quick phase transfer between anatase titanium dioxide and rutile titanium dioxide. The quick phase transfer and elevated barrier allows for a long fluorescence lifetime and better efficiency, boosting hydrogen peroxide production using visible and near infrared light. Researchers suspect that the graphene/rutile titanium dioxide can be reused for at least six cycles of standard reactions, making it even more efficient for producing hydrogen peroxide.
As for what's next, researchers are looking ahead to how to simplify the process. "In the follow-up work, we hope to develop simpler strategies to optimize the heterostructure of photocatalysis to further improve the utilization of photogenerated hot electrons. This photocatalytic system driven by photogenerated hot electrons on cheap noble-metal-free heterojunctions shows significant potential as a new artificial photosynthesis system," said Li.
More information: Weiyao Hu et al, Rapidly and mildly transferring anatase phase of graphene-activated TiO2 to rutile with elevated Schottky barrier: Facilitating interfacial hot electron injection for Vis-NIR driven photocatalysis, Nano Research (2022). DOI: 10.1007/s12274-022-4624-8
Journal information: Nano Research
Provided by Tsinghua University Press