Supercomputing and neutrons crack code to uranium compound's signature vibes

Oak Ridge National Laboratory researchers used the nation's fastest supercomputer to map the molecular vibrations of an important but little-studied uranium compound produced during the nuclear fuel cycle for results that could lead to a cleaner, safer world.
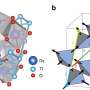
The study by researchers from ORNL, Savannah River National Laboratory and the Colorado School of Mines used simulations conducted on ORNL's Summit supercomputer and state-of-the-art neutron spectroscopy experiments conducted at the Spallation Neutron Source to identify key spectral features of uranium tetrafluoride hydrate, or UFH, a little-studied byproduct of the nuclear fuel cycle. The findings may enable better detection of this environmental pollutant and better understanding of how environmental conditions influence the chemical behavior of fuel cycle materials.
"In this kind of work, we don't have the luxury of choosing what kinds of materials we work with," said Andrew Miskowiec, an ORNL physicist and lead author of the study, published in The Journal of Physical Chemistry C. "We're often dealing with small quantities or even just particles of byproducts and degraded material that no one intended to make of compounds that we don't know much about. We need to know: If we found this material in the field, how would we recognize it?"
UFH forms when uranium tetrafluoride, a radioactive salt routinely used in producing uranium metal, begins to break down after immersion in water for 12 hours or longer. Even though scientists have studied uranium and its power to split the atom for nearly a century, most of those studies have focused on intentional results rather than unintended byproducts like UFH.
"From World War II through the Cold War, we have decades of study, but the main concern was making things work from a production standpoint, like building bombs and powering reactors," Miskowiec said. "UFH wasn't considered valuable for those purposes. That means it hasn't been studied as closely and isn't as well understood. We need to know as much as we can about these materials in order to know what to look for when we discover them in the wild."
Each of uranium's various molecular forms undergoes a unique set of vibrations, created by the dynamic motion of its atoms, that can act as a signature if scientists know what to look for. The research team used VISION, the world's highest-resolution inelastic neutron scattering spectrometer at the SNS, to bombard samples with neutrons, monitor the energy lost or gained, and capture the full range of UFH's vibrations.
"For other common characterization techniques, we would have had to dissolve or otherwise destroy the sample to study it," said Ashley Shields, an ORNL computational chemist and co-author of the study. "If we don't have a big sample to start with, we definitely don't want to destroy it before extracting as much information as possible. Spectroscopy gives us a way to gather data and preserve the sample for further analysis."
Conventional scattering methods rely on photons or electrons, which interact with an atom's outer shell and capture only a limited portion of the wide range of vibrations between atoms in a uranium compound. That's not a problem for neutrons, which penetrate all the way to an atom's nucleus.
"Neutrons are sensitive to all the atoms in the compound's structure, so we get the entire vibrational spectrum," Miskowiec said. "These extraordinary instruments at SNS gave us a huge amount of data, and now we needed a way to interpret it."
The team received an allocation of time on Summit, the Oak Ridge Leadership Computing Facility's 200-petaflop IBM AC922 supercomputing system, via the U.S. Department of Energy's Advanced Scientific Computing Research's Leadership Computing Challenge. They used density functional theory, a quantum-mechanical approach to estimating materials' structure, to model UFH's properties.
The combination of detail captured by VISION and the interpretation of large-scale, highly accurate density functional theory calculations made possible by Summit yielded the first complete picture of UFH's full vibrational spectrum for new insights into the compound's atomic structure.
"These are extremely large, intricate structures with a lot of atoms constantly vibrating in all directions with very little symmetry," Shields said. "Every break in the symmetry requires more calculations, increasing the compute time required to determine the vibrational properties. These computations allow us to visualize what kinds of vibrations these are, what the motion looks like, which atoms are participating in and causing each vibration, and at what frequency."
The team used the data to compare the calculated vibrational spectrum to the experimental one measured at the SNS, allowing for atomic-level identification of spectral features in the experimental data. The study required more than 115,000 node hours to render the results.
"Without Summit, these calculations couldn't have been done," Shields said. "There's a diversity of motion happening in the atomic structure we can tease out computationally that we just can't capture any other way."
Future studies will build on the findings to explore UFH's stability.
"We now have a better ability to identify this material in the field, and the results will be foundational for understanding other environmental aspects of the fuel cycle," Miskowiec said.
More information: Andrew Miskowiec et al, Inelastic Neutron Spectra of Uranium Tetrafluoride Hydrate, UF4(H2O)2.5, The Journal of Physical Chemistry C (2021). DOI: 10.1021/acs.jpcc.1c05747
Journal information: Journal of Physical Chemistry C
Provided by Oak Ridge National Laboratory