Learning from the single cell: A new technique to unravel gene regulation
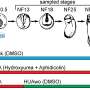
How is the activity of genes regulated by the packaging of DNA? To answer this question, a technique to measure both gene expression and DNA packaging at the same time was developed by Franka Rang and Kim de Luca, researchers from the group of Jop Kind (group leader at the Hubrecht Institute and Oncode Investigator). This method, EpiDamID, determines the location of modified proteins around which the DNA is wrapped. It is important to gather information about these modifications because they influence the accessibility of DNA, thereby affecting the gene activity. EpiDamID is therefore valuable for research into the early development of organisms. The results of the study are published in Molecular Cell.
In order to fit DNA into the nucleus of a cell, it is tightly packed around nuclear proteins called histones. Depending on the tightness of this winding, the DNA can be (in)accessible to other proteins. This therefore determines whether the process of gene expression, translation of DNA into RNA and eventually into proteins, can take place.
DNA packaging determine gene activity
The tightness of DNA winding around histones is regulated by the addition of molecular groups, so-called post-translational modifications (PTMs), to the histones. For example, if certain molecules are added to the histones, the DNA winding is loosened. This makes the DNA more accessible for certain proteins and causes the genes in this part of the DNA to become active, or expressed. Furthermore, proteins that are crucial for gene expression can directly recognize and bind the PTMs. This enables transcription: the process of DNA copying.
The regulation of gene expression, for instance through PTMs, is also known as epigenetic regulation. Since all cells in a body have the same DNA, regulation of gene expression is needed to (de)activate specific functions in individual cells. For instance, heart muscle cells have different functions than skin cells and so require different genes to be expressed.
Analysis of single cells using EpiDamID
To understand how PTMs affect gene expression, first authors Franka Rang and Kim de Luca designed a new method to determine the location of the modifications. Using this approach, called EpiDamID, researchers can analyze single cells, whereas previous methods were only able to measure a large group of cells. Analysis on such a small scale results in knowledge on how DNA winding differs per cell, rather than information on the average DNA winding of many cells.
EpiDamID is based on DamID, a technique which is used to determine the binding location of certain DNA-binding proteins. Using EpiDamID, the binding location of specific PTMs on histone proteins can be detected in single cells. Compared to others, a great advantage of this technique is that researchers need very limited material. Furthermore, EpiDamID can be used in combination with other methods, such as microscopy, to study regulation of gene expression on different levels.
Future prospects
Following the development of this technique, the Kind group will focus on the role of PTMs from the point of view of developmental biology. Because single cells are analyzed using EpiDamID, only a limited amount of material is needed to generate enough data. This allows researchers to study the early development of organisms from its first cell divisions, when the embryo consists of only a few cells.
More information: Jop Kind, Single-cell profiling of transcriptome and histone modifications with EpiDamID, Molecular Cell (2022). DOI: 10.1016/j.molcel.2022.03.009. www.cell.com/molecular-cell/fu … 1097-2765(22)00218-0
Journal information: Molecular Cell
Provided by Hubrecht Institute