Researchers discover a way to control cells in charge of tissue regeneration
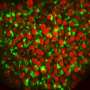
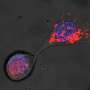
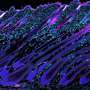
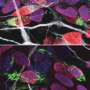
Tissue stem cells renew tissues by producing daughter cells that either remain as stem cells or differentiate into specialized cells, such as intestinal or skin cells. Certain stem cells seal the fate of their daughter cells already at the time of division. Researchers at the University of Helsinki have discovered how stem cells control the function of their daughter cells in cell division.
When a stem cell transfers its more active old mitochondria into one daughter cell, that cell loses its capacity to self-renew and is differentiated into a cell that maintains tissue function. The other daughter cell, receiving younger mitochondria, continues its life as a stem cell and remains capable of repairing tissue over and over again. The researchers found that the older mitochondria inherited by the differentiated daughter cell are better at maintaining metabolism based on cellular respiration.
"Switching on genes central to cellular functions ultimately determines the identity of cells, but metabolism appears to be the very first determinant of their fate," says Associate Professor Pekka Katajisto from the University of Helsinki's Helsinki Institute of Life Science (HiLIFE).
"When we restricted cellular respiration by reducing the quantity of a single protein enriched in the old mitochondria, the daughter cells actually remained self-renewing stem cells," says Doctoral Researcher Julia Govenius (née Döhla) from the University of Helsinki.
This demonstrates that small differences in mitochondrial quality can affect whole cell metabolism and, ultimately, the cell's fate in tissues.
"The self-renewal of stem cells can be influenced through surprisingly small and correctly timed metabolic changes," says Emilia Kuuluvainen, research coordinator of the Centre of Excellence in Stem Cell Metabolism. This finding is part of basic research that lays the groundwork for developing novel ways to promote tissue regeneration in, for example, degenerative diseases or accelerate recovery from tissue damage.
More information: Julia Döhla et al, Metabolic determination of cell fate through selective inheritance of mitochondria, Nature Cell Biology (2022). DOI: 10.1038/s41556-021-00837-0
Journal information: Nature Cell Biology
Provided by University of Helsinki