Hydrogel composite developed to help protective gear rapidly degrade toxic nerve agents

Scientists at Northwestern University in Evanston, Illinois have developed a hydrogel integrated with zirconium-based robust metal-organic frameworks (MOFs) that rapidly degrades organophosphate-based nerve agents used in chemical warfare. Unlike existing powdered MOF adsorbents, this hydrogel composite does not require added water and may be easily scaled up for use in protective masks or clothing. The work appears July 14 in the journal Chem Catalysis.
"Organophosphate-based nerve agents are among the most toxic chemicals known to humanity," says senior author Omar Farha, a professor of chemistry at Northwestern University. "Their use in recent global conflicts reflects the urgent need for personal protective gear, as well as the bulk destruction of chemical weapon stockpiles. In this work, we integrate MOFs and amine-containing cross-linked hydrogel into cloth to build a proper microenvironment to facilitate the fast degradation of nerve agents and supply real-time protection."
While MOFs have previously demonstrated an exceptionally fast ability to break down organophosphorus agents and chemicals that simulate them in the lab, these powdered adsorbents have proven difficult to directly integrate into protective cloths. When the nerve agents bind to their zirconium-6 clusters, they often deactivate the powder and fibrous composite catalysts. This pitfall calls for the use of alkaline solutions to regenerate the MOFs' catalytic sites—a requirement that does not prevent such MOFs from being used to eliminate stockpiled chemical weapons but which does impede their use in wearable protective gear.
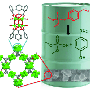
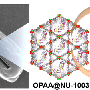
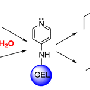
To overcome this challenge, Farha and colleagues designed a MOF-based textile composite system that uses water in an amine-based hydrogel to break down nerve agents. The material works by bringing together three key components for the hydrolysis reactions that dismantle the toxic organophosphorus agents. The zirconium node of the MOF provides a Lewis acidic site that activates the phosphorus center (the active part of the nerve agent), while hydrogel pores trap the necessary water. Basic amine groups in the hydrogel backbone generate hydroxyl groups to facilitate the nucleophilic attack on the organophosphorus substrate and subsequent displacement of the hydrolysis product on the zirconium center (i.e., catalytic turnover).
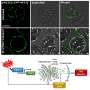
The researchers integrated this hydrogel composite with cotton fibers and tested it by applying a small aliquot of either a simulant or an actual nerve agent (tested in collaboration with the US Army Lab) to its surface. Next, they analyzed the product and substrate using nuclear magnetic resonance spectroscopy. They found that the composite chemically converted 99% of the agent within just 10 minutes, maintaining this high level of catalytic activity even after it was stored in a sealed vial for 3 months.
"The composite material developed here represents a significant improvement compared to what we previously developed," says Farha. "It is also important to note that the reactivities reported here with the composite in its solid state are comparable to those obtained in alkaline aqueous solutions."
Since the authors envision the novel hydrogel composite being used as a reactive layer in suits and masks, they note that further engineering and testing will be necessary to integrate it into these existing products. However, since the method used to produce the composite is simple and easily scalable, Farha suggests that large-scale production of MOF-based masks and suits may be possible in the future.
"We are in the process of optimizing the composite material to be suitable for real-world conditions," says Farha. "We hope in the future these materials will be commercialized and used to protect human life."
More information: Chem Catalysis, Ma et al.: "Near-instantaneous catalytic hydrolysis of organophosphorus nerve agents with zirconium-based metal-organic-framework hydrogel composites" www.cell.com/chem-catalysis/fu … 2667-1093(21)00091-9 , DOI: 10.1016/j.checat.2021.06.008
Provided by Cell Press