COVID-19 variants develop better lock-picking skills to invade human cells
Like expert lock pickers, COVID-19 variants may be more adept at breaking into and infecting human cells, according to new research conducted by FIU physicists.
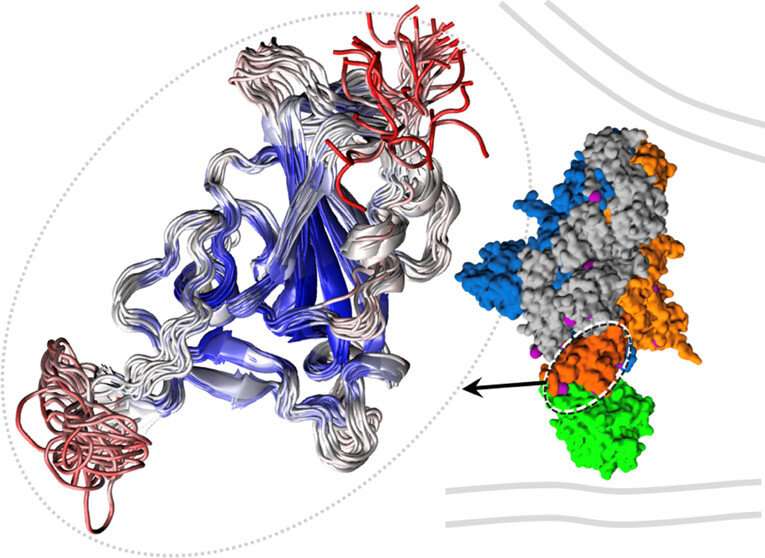
The variants are able to do this by flexing a spike protein that works like a lock pick, unlocking and slipping into a cell for infection. The better the virus can manipulate the spike protein, the easier time it has accessing the cell and eventually spreading in an unvaccinated population.
"Think of it as a moldable key," said physicist Prem Chapagain, the associate director of FIU's Biomolecular Sciences Institute and one of the study's authors. "The key itself is flexible and it finds the right groove and fits better because of that flexibility."
In computer simulations conducted at FIU, Chapagain, physics professor Bernard Gerstman and graduate students Nisha Bhattarai and Prabin Baral saw how the tip of the original coronavirus' spike protein wiggled but the tip of the South African variant wiggled even more. That extra flexibility, researchers said, helps it to better bind with human cells, though that alone may not fully account for the South African variant's higher infection rate.
These spike proteins are of particular interest to researchers, too, because some of the most effective coronavirus vaccines block the spike protein from working and infecting cells. While other studies have shown vaccines are effective against these variants, FIU researchers suggest more research is needed to determine whether changes seen in these and any future variants might make vaccines ineffective. Vaccines are still the best chance we have to cut the spread of the virus.
"If you can contain it everywhere and vaccinate most of the world's population, you don't have the breeding ground for the virus," Chapagain said. "We cannot give the virus a chance to acquire dangerous mutations. The more it spreads the more it mutates."
The study was supported by a National Science Foundation award and published in the Journal of Physical Chemistry B.
More information: Nisha Bhattarai et al, Structural and Dynamical Differences in the Spike Protein RBD in the SARS-CoV-2 Variants B.1.1.7 and B.1.351, The Journal of Physical Chemistry B (2021). DOI: 10.1021/acs.jpcb.1c01626
Journal information: Journal of Physical Chemistry B
Provided by Florida International University