Why frogs can't regenerate lost limbs like axolotls

In Lake Xochimilco of central Mexico dwells a rare salamander, the axolotl (Ambystoma mexicanum). In the wild, the axolotls do not metamorphose: adults very much resemble their larval counterparts and keep the external gills that ornate their head. Throughout their lives, axolotls have the ability to regrow lost parts of their body, such as a limb munched by a predator. The process may take months, but when it is complete, a regenerated leg is indistinguishable from the original: fully functional and scar-free. While this phenomenon is common in salamanders, it is rare in other vertebrates.
On the other side of the Atlantic, in the wetlands of Sub-Saharan Africa, lives the African clawed frog (Xenopus laevis), an animal to which developmental biologists have taken a liking. In contrast to the axolotl,Xenopusgoes through the process of metamorphosis, during which a tadpole turns into a frog and loses much of its ability to regenerate. When the leg of an adult is amputated, it is gradually replaced by a pointy 'spike' of unsegmented cartilage, giving the frog's leg a misleading Wolverine look.
Puzzled by this marked difference between axolotls and frogs, an international team of scientists led by the labs of Elly Tanaka at the Research Institute of Molecular Pathology (IMP) and Barbara Treutlein at ETH Zurich set out to investigate the process at the cellular and molecular levels. Their study, now published in the journal Developmental Cell, lays a new stone for our understanding of regeneration in vertebrates.
Blame the blastema
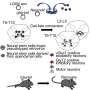
When our body gets hurt, various cells migrate to the wound to build a scar—the original tissues are lost, but the body can go on functioning. When an axolotl loses a limb, the same types of cells come together to form the blastema, a lump of dedifferentiated cells that rebuild the missing limb in all its complexity. From tendons to bone, cartilage, and ligaments, the stem cells that originate in the blastema allow all tissues to grow back.
"InXenopus, we found that the blastema cannot make the regenerative stem cells that would be necessary to regrow a normal limb," explains Tobias Gerber, currently a postdoctoral fellow at the European Molecular Biology Laboratory in Heidelberg, Germany. "Axolotls seem to have something extra that makes regeneration successful."
There are two possible reasons behind the blastema's strange behavior in the frog: the internal machinery of cells may prevent them from going back to a stem cell stage, or something in the frog's body, such as its immune system, may be at fault.
"We wanted to understand what blastema cells from adultXenopuscan do once they're decoupled from their environment. We transplanted cells from the blastema into the developing limb bud of tadpoles, where they should be unconstrained to contribute to limb development." says Tzi-Yang Lin, a key contributor to the study. "Our results clearly show that, even in this permissive embryonic environment, the adult blastema is unable to generate a functional limb."
Molecular reboot [not responding]
The logical next step was to explore the molecular basis of this difference. Using single-cell RNA sequencing methods, the scientists determined which genes were expressed differently in the cells of the adult frog blastema, the embryonic frog limb bud, and the axolotl blastema.
They found that the regeneration of the adult frog spike is molecularly distinct from its embryonic counterpart—as well as from the axolotl's. The scientists established a long list of genes that are either over- or underexpressed in the frog blastema—their next research move will be to find out what these genes do.
Even after the adult blastema cells were transplanted into the limb bud of a tadpole, their gene expression could not catch up. The cells remained unresponsive and did not contribute to forming a limb, revealing their intrinsic inability to be reprogrammed. The study could have important implications for regenerative medicine, as it provides new insights into the molecular basis of regeneration.
"The first question that comes up when I talk about my research on axolotl regeneration is always 'why can't humans regenerate a limb?'" says Yuka Taniguchi-Sugiura, senior research assistant in the Tanaka Lab. "We're using the African clawed frog as a stepping stone to understand why regeneration is so limited in other animals such as humans."
"Our study is the first to test the cells of the axolotl and Xenopusto to see if they can form regenerative stem cells in the same way. Eventually, we hope that our work will help our understanding of regeneration—or the lack thereof—in mammals," says Elly Tanaka, senior scientist at the IMP.
More information: Tzi-Yang Lin et al, Fibroblast dedifferentiation as a determinant of successful regeneration, Developmental Cell (2021). DOI: 10.1016/j.devcel.2021.04.016
Journal information: Developmental Cell
Provided by Research Institute of Molecular Pathology