How oxygen-producing cyanobacteria facilitated complex life

The "Great Oxygenation Event" (GOE), the process whereby the Earth's atmosphere was continuously enriched with oxygen, a waste product of photosynthesis, began ~2.43 billion years ago. The source, according to science, was photosynthesizing cyanobacteria. But why did this all-important turnaround occur so late? Cyanobacterial life existed, as rock samples show, at least 300 million years before the GOE. Achim Herrmann, who is researching the spread of early cyanobacteria in his doctoral thesis at TU Kaiserslautern, is hot on the trail for answers. His current research paper has now been published in the journal Nature Communications.
"There are many scientific theories that intertwine to explain why the proliferation of cyanobacteria required for the GOE was delayed," explains Herrmann, who is working on his doctorate with Michelle Gehringer in Geomicrobiology. "For example, they may have originated in fresh water, which covered then, as now, only a fraction of Earth's surface. It wasn't until they adapted to saltier waters and finally inhabited the open ocean that they were able to form sufficient amounts of biomass to cause a global change in Earth's atmosphere." Another theory is that the iron-rich ocean water may have initially been toxic to the photosynthesizing bacteria. Iron had accumulated in the marine environment predominantly in the form of highly soluble, reduced iron(II) ions during the Earth's then oxygen free "Archean" age.
In his research Herrmann built upon the iron toxin hypothesis. "We wanted to check whether iron(II) inhibits not only modern Cyanobacteria but also more primitive, marine strains, specifically Pseudanabaena sp. PCC7367 and Synechococcus sp. PCC7336, in their growth and photosynthetic activity," said the biologist.
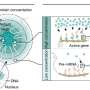
It quickly became apparent how crucial the experimental setup is. In already established systems where the bacteria are cultivated in closed glass bottles without oxygen, they demonstrated almost no growth: "The biological activity was very low in both strains, and almost completely suppressed in Synechococcus," Herrmann says. The solution: "A custom-built anaerobic workstation from the TUK metal workshop, in whose chambers the composition of the atmosphere can be regulated fully and automatically," he says. "Using this setup, we cultivated the cyanobacteria in large laboratory bottles with gas-permeable lids to allow gas exchange. The oxygen they produced was regularly removed from the system, and carbon dioxide was kept constant at proposed Archean atmospheric levels. Thus, we were able to realize a shallow marine oxygen oasis as implied in Archean rock samples."
As expected, the cyanobacteria "felt more comfortable" in the more authentic environment. But what happened when iron was injected in increasing concentrations? The bacteria from the Pseudanabaena strain grew consistently well, but more slowly than in the control system. In contrast, the Synechococcus strain clearly decreased its rate of cell division as iron increased. The oxygen produced primarily oxidized the dissolved Fe(II) ions instead of escaping into the atmosphere. And the oxygen production rate for both strains reached significantly higher values in the anoxically adjusted experimental environment than in the control setup with an oxygenated atmosphere, like that which surrounds us today. This would suggest that modern day atmospheric oxygen levels impair photosynthesis when compared to the anoxic atmosphere of Earth's past.
In addition, the formation of green rust, a mix of Fe(II) and oxidized iron Fe(III), was shown only in the culture system developed by Herrmann. The formation of green rust was accompanied by a strong decrease of biological activity, probably caused by iron oxides encrusting the bacterial cells. During the Archean, the formation of such green rust may have contributed decisively to banded iron formations, the most important source of iron ore today.
Finally, Herrmann changed the experimental scenario once again and simulated iron conditions for a tidal zone. Iron was added at night, when oxygen concentrations dropped towards zero due to no photosynthetic activity. The result: growth slowed significantly in both strains, but never stopped completely. This indicates that an Archean oxygen oasis could also have tolerated the influx of iron-rich water during the night. Here, too, the formation of green rust occurred, but could be further oxidized quickly and thus did not bring growth to a standstill.
All in all, Herrmann's research has filled in more gaps in the puzzle of Earth's history. He was able to illustrate for both cyanobacterial strains how the iron cycle might have proceeded in an Archean oxygen oasis, and that smaller colonized areas would probably have been sufficient for the start of the GOE due to the higher oxygen production rates. He has also developed a concept for growing cyanobacteria that better represents Archaean living conditions.
"I hope that with my research paper, I can help us better understand how our oxygen rich atmosphere was able to evolve in the first place," Herrmann says.
More information: A. J. Herrmann et al. Diurnal Fe(II)/Fe(III) cycling and enhanced O2 production in a simulated Archean marine oxygen oasis, Nature Communications (2021). DOI: 10.1038/s41467-021-22258-1
Journal information: Nature Communications
Provided by Technische Universität Kaiserslautern