This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Genes spatially organize for efficient mRNA splicing, study shows
The nucleus of each of your cells contains all the genetic information (the genome) necessary to build every type of cell and protein in your entire body. Like a complex library in a tiny space 50 times smaller than the width of a human hair, genes are organized into precise regions in three dimensions.
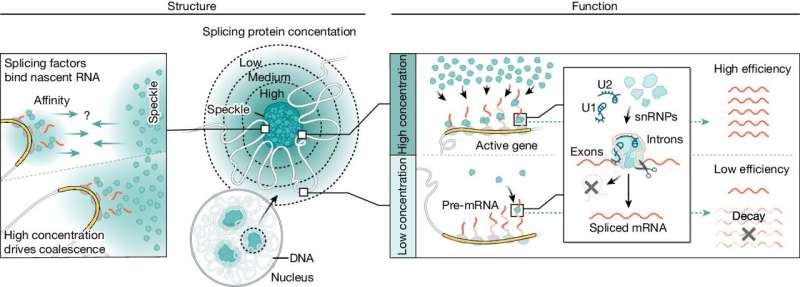
In addition to the genome, the nucleus contains structures called nuclear bodies that contain high concentrations of specific proteins and nucleic acids. The role of nuclear bodies, however, has been a mystery for nearly a century. A new study from the laboratory of Caltech's Mitch Guttman, professor of biology, shows that these nuclear bodies can act like miniature factories to enable efficient production of mRNA.
mRNA splicing is a fundamental part of the process that occurs when encoded DNA instructions are turned into a functional protein. After a gene is transcribed from DNA to RNA, extraneous sections (called introns) must be cut out, a process facilitated by splicing enzymes.
In the new study, the Caltech researchers examined a particular type of nuclear body, called the nuclear speckle, which contains high concentrations of splicing enzymes. Guttman's team discovered that the genome physically shifts itself around so that highly transcribed genes are in close proximity to speckles, enabling more efficient splicing.
The findings have implications for both fundamental scientific understanding of the relationship between nuclear structure and cellular function and, ultimately, for future therapeutics development for diseases in which mRNA splicing is affected, including for neurodegenerative conditions like ALS.
The paper describing the findings was published in the journal Nature on May 8. It is titled "Genome organization around nuclear speckles drives mRNA splicing efficiency."
For a decade, researchers in the Guttman laboratory have studied how the nucleus is spatially organized—the layout of the library, in other words. The 3D structures into which DNA is ordered make certain genes more or less accessible to the machinery that convert DNA into mRNA, and the new study shows that the physical structure of the genome links transcription with the process of splicing.
A muscle cell, for example, will shift its genome around so that highly transcribed genes for muscle activity are in physical proximity to nuclear speckles, where high concentrations of splicing enzymes make RNA splicing particularly efficient. A neuronal cell, on the other hand, will reorient its genome in space so that the genes necessary to produce cells specialized for neurological function are closer to the speckles.
"We know that splicing is dependent on enzymes, and any mutation that disrupts the concentration of enzymes has dramatic effects," says Prashant Bhat, a postdoctoral scholar research associate in biology and biological engineering and the study's first author.
"Previously, we tended to think of splicing as a constant—you make a pre-mRNA; you splice it. Now we know that the physical organization of the nucleus makes a big difference in the efficiency of splicing and, by extension, other processes."
Many diseases, including neurodegenerative disorders and cancer, have a common characteristic: dysfunctional RNA splicing. While it is not known whether improper splicing is a primary cause of disease or an effect, ultimately, the splicing process is a promising target for therapeutics.
For example, diseases in which an inadequate number of proteins are made (known as haploinsufficiency diseases) might be treated by repositioning the normal copy of the gene for that protein near nuclear speckles to boost splicing and restore healthy protein levels.
More information: Prashant Bhat et al, Genome organization around nuclear speckles drives mRNA splicing efficiency, Nature (2024). DOI: 10.1038/s41586-024-07429-6
Journal information: Nature
Provided by California Institute of Technology