Death enables complexity in chemical evolution
Simple systems can reproduce faster than complex ones. So, how can the complexity of life have arisen from simple chemical beginnings? Starting with a simple system of self-replicating fibers, chemists at the University of Groningen have discovered that upon introducing a molecule that attacks the replicators, the more complex structures have an advantage. This system shows the way forward in elucidating how life can originate from lifeless matter. The results were published on 10 March in the journal Angewandte Chemie.
The road to answering the question of how life originated is guarded by Spiegelman's monster, named after the American molecular biologist Sol Spiegelman, who some 55 years ago described the tendency of replicators to become smaller when they were allowed to evolve. "Complexity is a disadvantage during replication, so how did the complexity of life evolve?" asked Sijbren Otto, Professor of Systems Chemistry at the University of Groningen. He previously developed a self-replicating system in which self-replication produces fibers from simple building blocks and, now, he has found a way to beat the monster.
Death
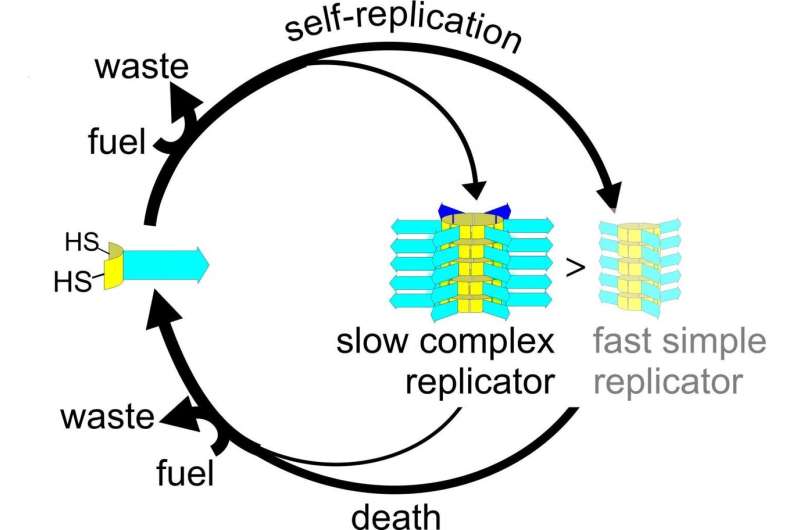
"To achieve this, we introduced death into our system," Otto explains. His fibers are made up of stacked rings that are self-assembled from single building blocks. The number of building blocks in a ring can vary, but stacks always contain rings of the same size. Otto and his team tweaked the system in such a way that rings of two different sizes were created, containing either three or six building blocks.
Under normal circumstances, fibers that are made up of small rings will outgrow the fibers with larger rings. "However, when we added a compound that breaks up rings inside the fibers, we found that the bigger rings were more resistant. This means that the more complex fibers will dominate, despite the smaller rings replicating faster. Fibers that are made from small rings are more easily "killed."'
Experiments
Otto acknowledges that the difference in complexity between the two types of fibers is small. "We did find that the fibers from the larger rings were better catalysts for the benchmark retro-aldol reaction than the simpler fibers that are made from rings with three building blocks. But then again, this reaction doesn't benefit the fibers." However, the added complexity protects the fibers from destruction, probably by shielding the sulfur-sulfur bonds that link the building blocks into rings.
"All in all, we have now shown that it is possible to beat Spiegelman's monster," says Otto. "We did this in a particular way, by introducing chemical destruction, but there may be other routes. For us, the next step is to find out how much complexity we can create in this manner." His team is now working on a way to automate the reaction, which depends on a delicate balance between the processes of replication and destruction. "At the moment, it needs constant supervision and this limits the time that we can run it."
Variants
The new system is the first of its kind and opens a route to more complex chemical evolution. "In order to achieve real Darwinian evolution that leads to new things, we will need more complex systems with more than one building block," says Otto. The trick will be to design a system that allows for the right amount of variation. "When you have unlimited variation, the system won't go anywhere, it will just produce small amounts of all kinds of variants." In contrast, if there is very little variation, nothing really new will appear.
The results that were presented in the latest paper show that, starting from simple precursors, complexity can increase in the course of evolution. "This means that we can now see a way forward. But the journey to producing artificial life through chemical evolution is still a long one," says Otto. However, he has beaten the monster guarding the road to his destination.
More information: Shuo Yang et al, Chemical Fueling Enables Molecular Complexification of Self‐Replicators, Angewandte Chemie International Edition (2021). DOI: 10.1002/anie.202016196
Journal information: Angewandte Chemie , Angewandte Chemie International Edition
Provided by University of Groningen