Bacteria adapt syringe apparatus to changing conditions
Some of the best-known human pathogens—from the plague bacterium Yersinia pestis to the diarrhea pathogen Salmonella—use a tiny hypodermic needle to inject disease-causing proteins into their host's cells, thereby manipulating them. This needle is part of the so-called type III secretion system (T3SS), without which most of these pathogens cannot replicate in the body.
Only recently it was discovered that large parts of the T3SS are not firmly anchored to the main part of the system, but are constantly exchanging during function. However, the significance of this phenomenon remained unclear. Researchers in the laboratory of Andreas Diepold at the Max Planck Institute for Terrestrial Microbiology have now discovered that this dynamic behavior allows the bacteria to quickly adapt the structure and function of the injection apparatus to external conditions.
The digestive system: A sea change for bacteria
Human digestion starts with a neutral to slightly alkaline environment in the mouth and esophagus, which the addition of gastric acids suddenly changes to strongly acidic in the stomach—an environment that many pathogens do not survive. The actual target of Yersinia enterocolitica, the pathogenic bacteria investigated in the study, is the intestine. Here, pH-neutral conditions are restored.
But how do the bacteria manage to adapt so quickly to the changing conditions, and how is this controlled? Ph.D. student Stephan Wimmi, the first author of the study, was able to demonstrate that a protein in the bacteria's membrane acts as a sensor for the pH value. In a collaboration with Ulrike Endesfelder's lab at the Max Planck Institute, he found that this protein becomes more motile at low (= acidic) pH and thus transmits the signal to the T3SS components inside the bacterium.
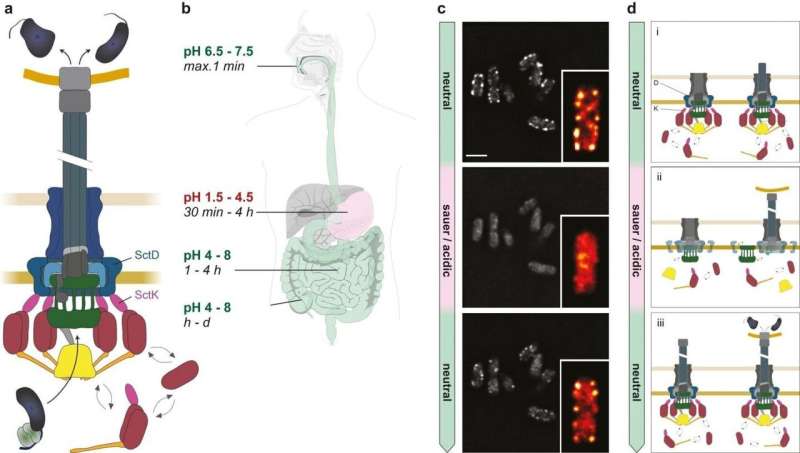
Flexibility prevents misfiring
In an acidic environment like the stomach, the mobile components do not bind to the rest of the apparatus (including the needle itself), so that the injection system remains inactive. As soon as the bacteria enter a pH-neutral environment—as it is found in the intestine—the dynamic proteins reassemble, so that the T3SS can quickly become active at these sites—to the possible distress of the infected person.
The researchers speculate that the newly discovered effect may allow the bacteria to prevent an energy-consuming 'misfiring' of the secretion system in the wrong environment, which could even activate the host's immune response. On the other hand, the mobility and dynamics of the structure allows the system to be rapidly reassembled and activated under appropriate conditions.
Protein mobility and exchange are increasingly being discovered in complexes and nanomachines across all domains of life; however, the utility of these dynamics is mostly not understood. The new results from Marburg show how protein exchange allows to respond flexibly to external circumstances—an immense advantage, not only for bacteria.
More information: Stephan Wimmi et al, Dynamic relocalization of cytosolic type III secretion system components prevents premature protein secretion at low external pH, Nature Communications (2021). DOI: 10.1038/s41467-021-21863-4
Journal information: Nature Communications
Provided by Max Planck Society