Novel catalysts improve efficiency of urea synthesis at ambient conditions
Converting both nitrogen (N2) and carbon dioxide (CO2) into value-added urea molecules via C-N coupling reaction is a promising method to solve the problem of excessive CO2 emissions.
Compared with huge energy consumption industrial processes, electrochemical urea synthesis provides an appealing route under mild conditions. However, it still faces challenges of low catalytic activity and selectivity.
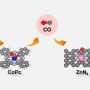
A research team led by Prof. Zhang Guangjin from the Institute of Process Engineering (IPE) of the Chinese Academy of Sciences fabricated Bi-BiVO4 Mott-Schottky heterostructure catalysts for efficient urea synthesis at ambient conditions.
This work was published in Angewandte Chemie International Edition on Feb. 25.
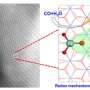
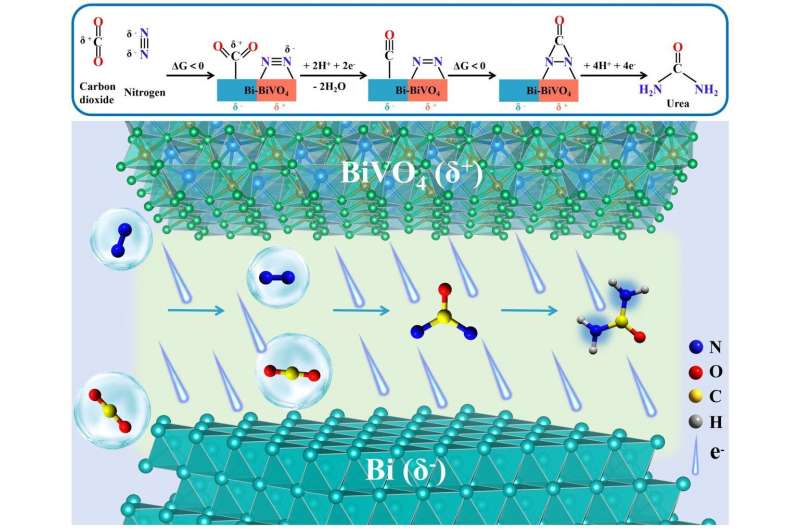
The spontaneous charge transfer at the heterointerfaces promotes the formation of space-charge region. "The space-charge region not only facilitates the targeted adsorption and activation of CO2 and N2 molecules on the generated electrophilic/nucleophilic regions, but also effectively suppresses CO poisoning and the formation of endothermic *NNH intermediate," said Prof. Zhang.
The adsorbed *N2 can promote CO2 reduction to form CO, and then the generated CO will further react with *N2 to produce the desirable *NCON* intermediate via electrochemical C-N coupling reaction.
"The subsequent protonation process preferentially undergoes the alternating mechanism until the formation of urea," said Prof. Zhang.
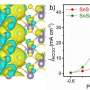
The researchers used linear sweep voltammetry (LSV) to preliminarily evaluate the potential performance of urea electrosynthesis with Bi-BiVO4 hybrids. The results showed that Bi-BiVO4 hybrids exhibited good performance in electrocatalytic nitrogen reduction reaction (NRR) and CO2 reduction reaction (CO2RR), which ensured the electrocatalytic production of urea.
More information: Menglei Yuan et al, Unveiling Electrochemical Urea Synthesis by Co‐Activation of CO2 and N2 with Mott‐Schottky Heterostructure Catalysts, Angewandte Chemie International Edition (2021). DOI: 10.1002/anie.202101275
Journal information: Angewandte Chemie International Edition
Provided by Chinese Academy of Sciences