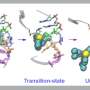
Supercomputer models describe chloride's role in corrosion
Researchers have been studying chloride's corrosive effects on various materials for decades. Now thanks to high-performance computers at the San Diego Supercomputer Center (SDSC) at UC San Diego and the Texas Advanced Computing Center (TACC), detailed models have been simulated to provide new insight on how chloride leads to corrosion on structrual metals, resulting in economic and environmental impacts.
Conducted by a team from Oregon State University's (OSU) College of Engineering, a study discussing this newfound information was published in Materials Degradation, a Nature partner journal.
"Steels are the most widely used structural metals in the world and their corrosion has severe economic, environmental, and social implications," said study co-author Burkan Isgor, an OSU civil and construction engineering professor. "Understanding the process of how protective passive films break down helps us custom design effective alloys and corrosion inhibitors that can increase the service life of structures that are exposed to chloride attacks."
Isgor worked closely with OSU School of Engineering colleague Líney Árnadóttir as well as graduate students Hossein DorMohammadi and Qin Pang on conducting the study. As a chemical engineering associate professor, Árnadóttir said her work often uses computational methods to study chemical processes on surfaces with applications in materials degradation.
"We frequently collaborate with experimental groups and use experimental surface science tools to complement our computational methods," she said. "For this study we relied on allocations from the National Science Foundation's (NSF) Extreme Science and Engineering Discovery Environment (XSEDE) so that we could use Comet and Stampede2 to combine different computational analyses and experiments applying fundamental physics and chemistry approaches to an applied problem with potentially great societal impact."
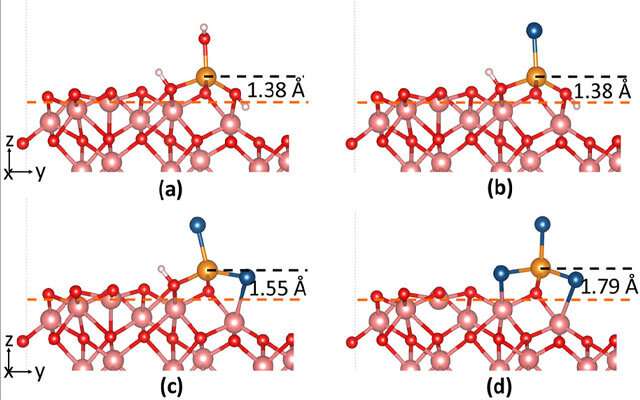
The OSU team used a method called density functional theory (DFT) to investigate the structural, magnetic, and electronic properties of the molecules involved. Their simulations were also corroborated by others using reactive molecular dynamics (Reax-FF MD), which allowed them to accurately model the chemistry-based nanoscale processes that lead to chloride-induced breakdown of iron passive films.
"Modeling degradation of oxide films in complex environments is computationally very expensive, and can be impractical even on a small local cluster," said Isgor. "Not only do Comet and Stampede2 make it possible to work on more complex, more realistic, and industrially relevant problems, but also these high-performance computers let us do so within a reasonable timeframe, moving knowledge forward."
More information: Qin Pang et al. Thermodynamic feasibility of the four-stage chloride-induced depassivation mechanism of iron, npj Materials Degradation (2020). DOI: 10.1038/s41529-020-00131-8
Journal information: Nature
Provided by University of California - San Diego