January 15, 2021 report
Intimate associations between SARS-CoV-2 and mitochondria suggest new angles of attack
As one wise pundit recently observed, "everybody is a virologist now." For the many people whose interest in biology formerly began and ended with "the mitochondria is the powerhouse of the cell," a second axiom can now be offered, namely, that the virus is the thief of power. In other words, what the mitochondria giveth, the virus taketh away.
It is only through the massive oxidative capabilities of mitochondria that cells of the immune system can generate enough energy within a sufficiently short period of time to power an effective immune response. This response includes massive short-order construction projects where cascading waves of signaling factors, antibodies and the armies of clones that pump them out are hastily hardscrabbled together. It is this same power that a virus hijacks upon gaining entry to a cell to use for copying, transcribing and translating their genomes (not always in that specific order) to almost exponentially replicate and propel themselves through the body at large.
It should therefore be no surprise that mitochondria and viruses are, at least in a molecular sense, quite well aware of each other. For example, it has been shown that the Orf9b accessory protein of SARS-CoV-2 interacts with the mitochondrial transport protein TOM70, while Orf9c interacts with respiratory complex I. The Nonstructural protein 2 (NSP2) has been localized to nuclear and mitochondrial prohibitins which in turn form a 16-20 subunit ring at the inner membrane. Prohibits are also believed to act as viral receptors for the Chikungunya and Dengue 2 viruses.
In a paper recently published in the journal Frontiers in Aging Neuroscience, researchers from Texas Tech University explore the idea that some viruses, including SARS-CoV-2, could even replicate within mitochondria-derived structures. This idea was initially established by Keshav K. Singh and colleagues who first began investigating potential hijacking of mitochondria by SARS-CoV-2. The authors say "mitochondria-derived" because in the absence of full dynamic imaging of double-membraned vesicle (DMV) formation within associated inclusions of mitochondria, endoplasmic reticulum (ER), golgi and virus, the necessary actions that seemingly must occur for the virus to complete its life cycle can only be inferred.
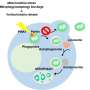
Mitochondria closely associated with the ER where they are embraced by external rings of contractile dynamin-related peptide (DRP1) molecules which squeeze them down to diameters small enough for spontaneous fusion and budding to occur. The authors note that in the original SARS-CoV-1, ORF-9b enhances mitochondrial fusion and reduces the levels of Drp1. Budding off DMVs packed with their own mtDNA nucleoids, which then fuse with the plasma membrane of the cell, is important business for mitochondria. Exporting these highly immunogenic lures are one way white blood cells sacrifice their own, in a sense, to ramp up immune responses. This all sounds a little familiar—during its lifecycle, SARS viruses must clothe their own genetic material in suitable double membrane form before beginning its transcellular journal.
In a another paper recently published in Scientific Reports, Pinchas Cohen and his team compared mitochondrial-COVID interactions to those of other viruses including respiratory syncytial virus, seasonal influenza A virus and human parainfluenza virus 3. One important finding was that in SARS-CoV-2, the levels of respiratory complex I components were reduced. Reduced complex I activity can also reduce levels of reactive oxygen species (ROS). The authors describe how host innate immunity is regulated by mitochondrial antiviral signaling proteins (MAVS). Under normal conditions, these MAVs interact with mitochondrial fusion factors like MFN2. However, after infection, mitochondria are tethered to the ER by MFN-2, whereupon the MAVS interacts with important kinases, namely, TANK binding kinase 1, IKKA, and IKKB.
Other new research shows that SARS-CoV-2 virus may go even further, suggesting that in peripheral blood mononuclear cells of patients with COVID-19, the virus deliberately manipulates the metabolic functions of mitochondria to their own advantage. In particular, the authors show increases in the mitokine FGF-21, and also increases in glycolysis. They propose that since FGF-21 correlates with disease severity, it could serve as a biomarker for COVID-19-related mitochondrial dysfunction. Since mitochondria play a key role in the initiation and development of cytokine storm, specific mitochondrial pathways in immune cells might be targeted clinically.
To get some more perspective, it is worth mentioning a few other important details regarding the SARS-CoV-2 genome. At around 30 kilobases long, it is twice the size of mtDNA, and over three times as long as the HIV genome. HIV is also a positive sense RNA virus; however, it is double-stranded, and integrates within the host cell genome. Although SARS-CoV-2 normally completes its life cycle in the cytoplasm, some recent evidence suggests that it, too, can be reverse-transcribed and integrated into nuclear DNA. While mtDNA is normally entirely circulatized (save perhaps in some heart muscle cells), SARS-CoV-2 can sometimes be circularized into circRNAs of many different sizes, although the implications of this are unknown. Like host nuclear DNA and mtDNA, the SARS-CoV-2 genome also contains unique G-quadruplex formations. These often enigmatic structural formations at specific guanine repeats are also potential therapeutic targets.
No SARS cabinet of curiosities would be complete without some ode to the still largely inexplicable furin cleavage site (FCS). While a few recombination theories have been bantered about, the actual mechanism is still an open question. For inspiration to answer this vexing question, we offer the charming and now famous genetics how-to video from the Cambridge iGEM Institute.
More information: Miller, B., Silverstein, A., Flores, M. et al. Host mitochondrial transcriptome response to SARS-CoV-2 in multiple cell models and clinical samples. Sci Rep 11, 3 (2021). doi.org/10.1038/s41598-020-79552-z
Saima Ajaz et al. Mitochondrial metabolic manipulation by SARS-CoV-2 in peripheral blood mononuclear cells of patients with COVID-19, American Journal of Physiology-Cell Physiology (2020). DOI: 10.1152/ajpcell.00426.2020
Riya Ganji et al. Impact of COVID-19 on Mitochondrial-Based Immunity in Aging and Age-Related Diseases, Frontiers in Aging Neuroscience (2021). DOI: 10.3389/fnagi.2020.614650
Journal information: Scientific Reports
© 2021 Science X Network