Rationally designing hierarchical zeolites for better diffusion and catalyst efficiency
Thanks to various crystalline topologies, tunable chemical composition, high (hydro)thermal stability, and controllable surface acidity/basicity, zeolites are widely used in petroleum refining, petrochemical manufacture, fine chemical synthesis, biomedicine, environmental chemistry, etc. However, for many zeolite-catalyzed reactions, the molecular diameters of the reaction species involved are often larger than the pore apertures of the zeolites. This leads to undesired diffusion resistance between the bulk phase and the active centers of the catalyst, thereby significantly reducing the catalyst efficiency.
Alleviating diffusion resistance and improving catalyst efficiency of the zeolite-based catalyst is always one of the most concerned issues in academia and industry. Within the past decades, tools for integrating hierarchical micro-/mesoporous structures into zeolites for better diffusion and catalyst efficiency have been greatly enriched.
However, in the real industrial catalysis processes, even if zeolitic component contains hierarchically porous structure, it is just one of the components of the multi-component industrial catalyst. The zeolite-based industrial catalyst is essentially hierarchical structure composed of microporous zeolitic and macroporous non-zeolitic components. When the hierarchically porous structure is integrated, the catalyst also has a micro-/meso-/macroporous trimodal hierarchical structure. Obviously, the hierarchical pore structure of industrial zeolite-based catalysts exists in two levels: 'inside the zeolitic component' and 'between the components of the industrial catalyst.'
In a new review paper published in the Beijing-based National Science Review, scientists at the China University of Petroleum in Qingdao, China (Peng Peng, Zi-Feng Yan), China National Petroleum Company in Beijing, China (Xiong-Hou Gao), and French National Center for Scientific Research (CNRS) in Caen, France (Svetlana Mintova) analyzed the state-of-the-arts in rational design of hierarchical micro-/mesoporous structures from catalytic reaction engineering point of view.
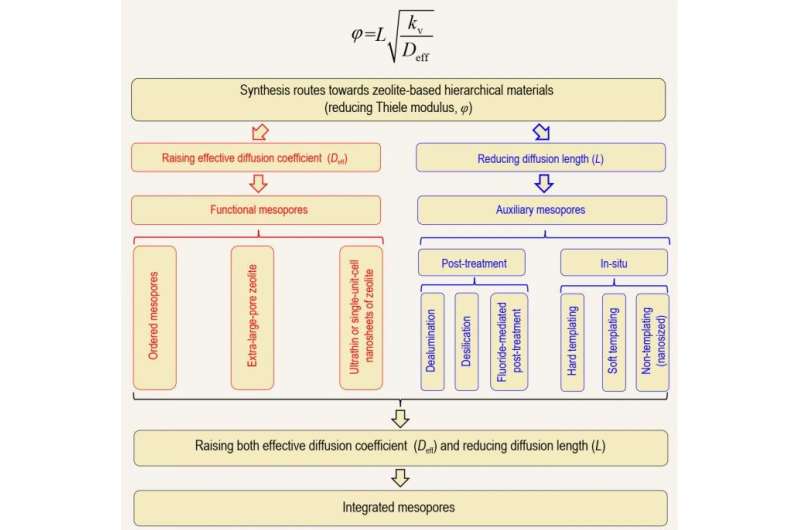
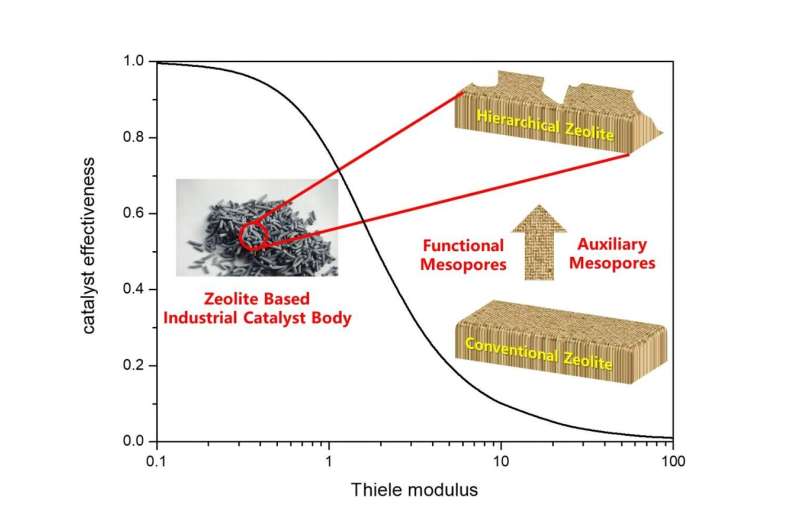
From the perspective of catalytic reaction engineering, the quantitative indicators for evaluating catalyst efficiency are catalyst effectiveness factor (η) and Thiele modulus (φ). If the catalyst system undergoes strong diffusion resistance (η<0.25), then η is the reciprocal of φ, so increased η means decreased φ. Based on the definition of φ, enhancing η can be achieved by either increasing the effective diffusion coefficient (Deff) or shortening the diffusion path (L). Based on this, the mesoporous structure in hierarchical zeolite can be divided into three types: (1) 'functional mesopores' (increase effective diffusion coefficient, Deff); (2) 'auxiliary mesopores' (shorten the diffusion path, L); and (3) 'integrated mesopores' (simultaneously increase Deff and shorten L). For hierarchical zeolite materials, excellent pore interconnectivity can ensure rapid diffusion and desorption of products formed on the active sites of the micropores, thereby avoiding deactivation. For a cascade reaction network like fluid catalytic cracking (FCC), well-designed hierarchically porous structure can ensure the interconnection between micro- and mesopores, which is very important for the reaction relay in FCC process.
Zeolite with a hierarchical porous structure is just one of the components of real industrial catalysts. In order to meet the requirements of mechanical strength, hydrothermal stability, resistance to poisoning and coking in the industrial catalytic processes, industrial catalysts need to add other non-zeolitic components. Although the interaction mechanism between the industrial catalyst components is not fully understood, the non-ideal matching of the porous structures between the zeolitic and the non-zeolite components can cause reducing performance of the hierarchical pore zeolite components. The coordination of pores interconnectivity of hierarchical zeolites and other non-zeolitic components in industrial catalysts is an urgent issue to be addressed prior the industrial applications of hierarchical zeolites.
The ultimate goal for preparing hierarchically porous material is to fully release its potential at industrial scale by controlling the hierarchical pore structure, different components' locations and interconnectivity that play a pivotal role on enhancing of their catalytic efficiency. Developing combined in-situ or operando spectroscopic, microscopic or diffraction techniques is the key to unravel the structure-activity relationship of hierarchical zeolites as a component in industrial catalysts.
More information: Peng Peng et al, Diffusion and catalyst efficiency in hierarchical zeolite catalysts, National Science Review (2020). DOI: 10.1093/nsr/nwaa184
Provided by Science China Press