Bimetallic catalyst helps to synthesize tunable imines and secondary amines
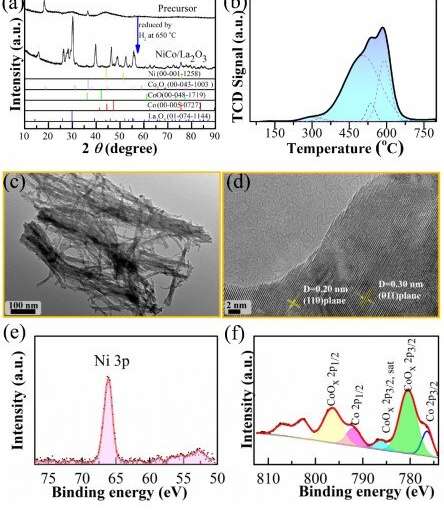
In a recent study, scientists from the Institute of Solid State Physics, Hefei Institutes of Physical Science prepared NiCo5 bi-metallic catalyst to activate carbon-nitrogen bond under mild reaction conditions.
Imines, amines and their derivatives have been regarded as very important collections of nitrogen-contained compounds in subsequent manufacturing of various organic chemicals. They were typically generated through a condensation reaction between primary-amine and aldehyde catalyzed by inseparable and expensive homogeneous catalysts. Therefore, it is highly desirable to develop green technologies to selectively produce imine and secondary-amine in one-pot.
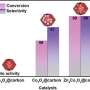
In this study, by constructing high-efficiency hydrogenation and coupling bifunctional catalysts, the selective synthesis of C=N double bond and C-N bond could be controllably achieved by simply adjusting reaction parameters, and a highly efficient bi-metallic catalyst NiCo5 was purposefully constructed to selectively synthesize imines or secondary-amines by facilely tuning of reaction parameters.
The synthesized catalyst was found to have dual-functional catalytic activity and adjustable hydrogenation performance was the key factor to achieve selectivity. With the introduction of another metal (Co), an interaction between the Ni and Co could be generated for further adjusting the activity and stability to obtain a suitable catalytic performance.

Further experimental and theoretical calculation results also proved that the interactions between Ni and Co as well as active metal and support were the key influencing factors of high selectivity.
This study paves way for designing efficiently selective catalysts in multi-step reactions.
More information: Chun Chen et al. Tunable synthesis of imines and secondary-amines from tandem hydrogenation-coupling of aromatic nitro and aldehyde over NiCo5 bi-metallic catalyst, Applied Catalysis B: Environmental (2020). DOI: 10.1016/j.apcatb.2020.119448
Journal information: Applied Catalysis B: Environmental
Provided by Chinese Academy of Sciences