Harvesting hydrogen from nanogardens
Easily produced, nature-like nanostructures of cobalt phosphide are highly effective catalysts for the electrolysis of water, according to research performed by chemist Ning Yan and his team at the University of Amsterdam's Van 't Hoff Institute for Molecular Sciences together with co-workers from the School of Physics and Technology at Wuhan University, China. In a paper featured on the front cover of the Journal of Materials Chemistry A, they describe how relatively straightforward electrochemical deposition methods yield grass-, leaf-, and flower-like nanostructures that carry the promise of efficient hydrogen generation.
For preparing nanostructures, top-down approaches such as lithography have since long been common. This has proven to be quite useful in semiconductor fabrication, but for more dedicated applications, it is time-consuming and not particularly cost-effective. As an alternative, many researchers explore the bottom-up synthesis of nanostructures, for instance, based on the self-assembly of molecules or nanoscale building blocks. However, achieving geometry control often requires costly additives and surfactants, rendering large-scale material preparation quite challenging.
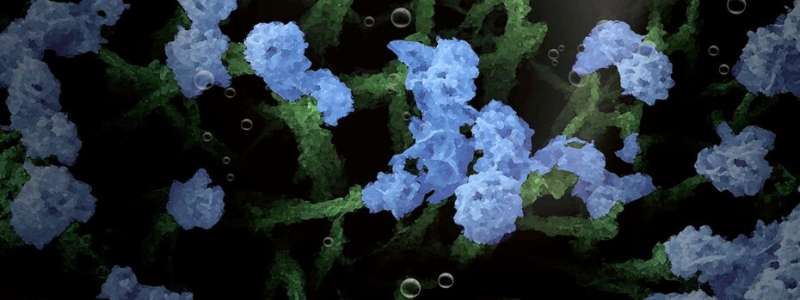
As an alternative, Assistant Professor Ning Yan, together with his Ph.D. students Jasper Biemolt and Pieter Laan of the University of Amsterdam's Van 't Hoff Institute for Molecular Sciences, have now explored a relatively straightforward method of electro-deposition of cobalt hydroxide. In cooperation with researchers at the School of Physics and Technology at Wuhan University, China, they have been able to design and prepare a variety of nano-architectures that resemble various items in a garden: soil, sprouts, grasses, flowers, and leaves.
The researchers report that they have mastered the system in such a way that they are able to grow any of these structures at will.
Adding to this, they were able to render the nanostructures catalytically active by a simple phosphidation procedure. The resulting cobalt phosphide nanostructures display bifunctional catalytic activity in electrolytic water splitting, enhancing both hydrogen and oxygen generation reactions.
Hierarchical nanostructures through controlled synthesis
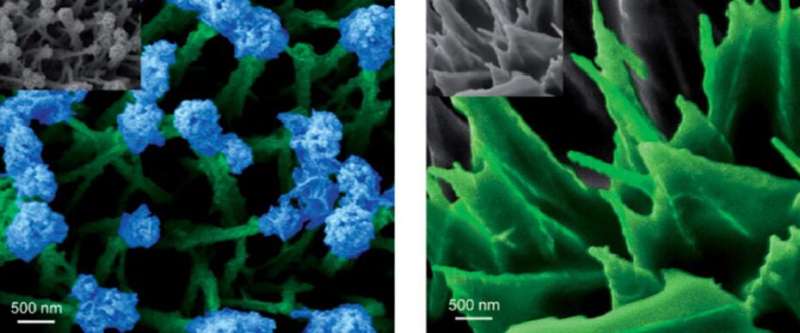
Ning Yan and co-workers grew their nanogardens on a cloth consisting of carbon fibers of around 10 micrometers in diameter, a common electrode material in the fuel cell and electrolyser industries. The gardening started with depositing a layer of "soil" by hydrothermally encapsulating the fibers with a dense layer of cobalt hydroxide. This layer increased the structural stability of the nanostructures. Through variation of the ion concentration and temperature, they were able to induce the "sprouting" of grass-like features that are strongly "rooted" in the soil.
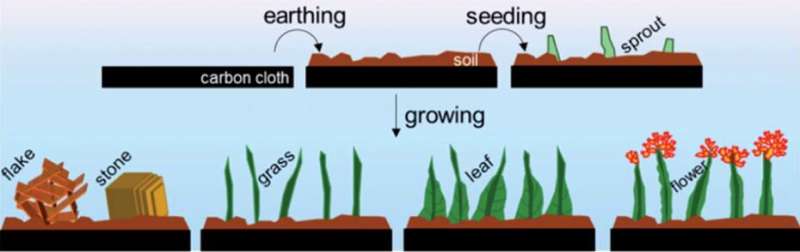
These grasses have an average length of 1.5 μm and a thickness of around 100 nm. To add blossoms and leaves to the grassy features, the researchers applied an electrodeposition method. In a diluted solution, electrodeposition dominantly proceeds from the tip of the grass stem, where the small radius of curvature results in a higher space charge density. In more concentrated solutions, the electrodeposition mainly proceeds from the bottom of the stems. This results in the deposition of "leafy" features, which in fact are interwoven dendritic deposit structures.
After converting the cobalt hydroxide nanostructures to cobalt phosphide by means of phosphidation, the researchers evaluated their catalytic activity in a setting that adequately represented industrially relevant conditions. As it turned out, the performance of the catalyst in an acidic environment is among the best of today's superior non-precious metal catalysts for hydrogen evolution. Furthermore, in acidic as well as alkaline and neutral conditions, the flowery nanofeatures resulted in significantly larger turnover frequencies than the leafy features, particularly at higher overpotentials when hydrogen evolution is influenced by mass transport limitations. The researchers attribute this to the geometry of the nanofeatures where the flowers enable smoother unloading of hydrogen. However, the different reaction environments at the top and bottom positions of the nanostructures complement each other, resulting in optimum overall performance.
Finally, in electrolysis experiments on water splitting, the researchers showed that their nanogardens not only catalyze the hydrogen evolution reaction but also the oxygen evolution. This bifunctional activity was shown using a symmetric two-electrode setup with completely identical nanogardens at the anode and cathode. The team will further investigate the use of electrons to control the growth of nanocrystals in an "electrified" materials synthesis that holds promise for a sustainable future.
More information: Xiaoyu Yan et al. "Nano-garden cultivation" for electrocatalysis: controlled synthesis of Nature-inspired hierarchical nanostructures, Journal of Materials Chemistry A (2020). DOI: 10.1039/d0ta00870b
Journal information: Journal of Materials Chemistry A
Provided by University of Amsterdam