Pushing the study of noncovalent intermolecular interactions into a new area
Rovibrational inelastic scattering of molecules has now been studied for many years. In so-called state-to-state experiments, the reagent molecules are prepared in a pure quantum state before the collision and the distribution of the scattered fragments over all energetically accessible states is detected, as well as the direction in which they fly apart. In a recent publication in Nature Chemistry, a research team within the Institute for Molecules and Materials of the Radboud University, together with several laboratories in the U.S., have collaborated to "push the study on noncovalent intermolecular interactions into a currently unprobed area."
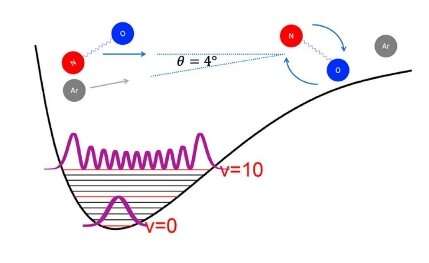
Most previous studies employed reagent molecules in their ground (v=0) vibrational state, which greatly simplifies the electronic structure and quantum dynamical scattering calculations that accompany the measurements. The aim was to study collisions between highly vibrationally excited NO molecules and atoms at different collision energies. "We wanted to study the effect of the vibrational motion of the NO-molecule on phenomena such as scattering resonances and diffraction oscillations," Matthieu Besemer, Ph.D. student in the Theoretical Chemistry and Spectroscopy of Cold Molecules Departments, explains. The research results have been published in Nature Chemistry journal, titled "State-to-state scattering of highly vibrationally excited NO at broadly tunable energies."
Strongly vibrating molecules
In the experimental and theoretical study of rotationally inelastic collisions between NO molecules and argon atoms, the experiments are exploring a new realm, by combining stimulated emission pumping to prepare the NO molecule in an arbitrary rovibrational state with a near- copropagating molecular beam geometry that allows collision energies below 1 kelvin. The NO molecule is initially excited to a high (v=10) vibrational state, which is a challenge for theory since the current knowledge of molecular interactions is mostly limited to the ground state.
Theoretical calculations
The theoretical results only agree with the measurements when the scattering calculations explicitly account for the vibrational motion of NO. "This work pushes the underlying calculations to the limit of what is possible at present. It is anticipated that further work in this direction will also require further extension of the available computational methods and, at the same time, teach us more about the interactions between molecules in their excited states," Besemer says. "Ultimately, this unique experimental technique will be used to study other systems as well."
The Spectroscopy of Cold Molecules group studies molecular collision processes at low energies and with unprecedented precision and the Theoretical Chemistry Department explains and predicts properties of molecules, clusters, and molecular solids. Both groups are part of IMM and have collaborated for many years resulting in numerous shared publications.
More information: Chandika Amarasinghe et al. State-to-state scattering of highly vibrationally excited NO at broadly tunable energies, Nature Chemistry (2020). DOI: 10.1038/s41557-020-0466-8
Journal information: Nature Chemistry