Accelerating biological systems design for sustainable biomanufacturing

Northwestern University synthetic biologists have developed a new rapid-prototyping system to accelerate the design of biological systems, reducing the time to produce sustainable biomanufacturing products from months to weeks.
As global challenges like climate change, population growth, and energy security intensify, the need for low-cost biofuels and bioproducts—like medicines and materials—produced using sustainable resources increases. Industrial biotechnology, which uses microbial cellular factories to harness enzyme sets that can convert molecules to desirable chemical product, has shown potential to address these needs. However, designing, building, and optimizing these pathways in cells remains complex and slow, unable to keep up with the dynamic shifts in needs.
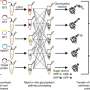
The new platform, called in vitro Prototyping and Rapid Optimization of Biosynthetic Enzymes (iPROBE), provides a quick and powerful design-build-test framework to discover optimal biosynthetic pathways for cellular metabolic engineering that could impact a range of industries (or issues) from clean energy to consumer products.
"For the first time, we show that cell-free platforms can inform and accelerate the design of industrial cellular systems," said Michael Jewett, Walter P. Murphy Professor of Chemical and Biological Engineering and Charles Deering McCormick Professor of Teaching Excellence at the McCormick School of Engineering, who directs Northwestern's Center for Synthetic Biology. "We accomplished in approximately two weeks what traditionally would have taken six to 12 months. Our findings will help accelerate the pace at which we can enable sustainable biomanufacturing practices."
The platform leverages Northwestern's leadership in cell-free synthetic biology and comes into play in three recently published studies, each led by Jewett.
"iPROBE stands to help scientists identify the best sets of enzymes for a variety of sustainable chemicals and bring them into manufacturing at scale," Jewett said. "We envision this cell-free system as an engine to help realize the future bioeconomy."
Adopting a cell-free approach
"In Vitro Prototyping and Rapid Optimization of Biosynthetic Enzymes for Cell Design," published June 15 in the journal Nature Chemical Biology, describes how iPROBE works.
To manufacture sustainable chemicals, synthetic biologists stitch together protein enzymes to carry out individual molecular transformations, converting readily available stock—like glucose or carbon dioxide—to a new product. Current testing methods require these enzymes get encoded in DNA, placed on a single plasmid molecule, and then inserted into a living cell. The process must be repeated each time to study a different set of enzymes in hopes of determining the most optimal grouping.
"The result is that the design cycles are just too slow," Jewett said. "We end up needing hundreds of combined person years of development to bring a product to market. That's too slow to address challenges like climate change and other rapidly growing problems we face."
iPROBE bypasses the limitations of engineering living organisms using cell-free protein synthesis to enrich biosynthetic enzymes in test tubes to carry out transformations. Combined with computational design algorithms developed by Lockheed Martin, the system rapidly studies pathway enzyme ratios, tuning individual enzymes in the context of the desired multi-step pathway, screening for high-performance enzymes, and discovering enzymes with optimal functionalities.
"iPROBE had to be multifaceted and easy to use," said Ashty Karim, first author on the paper and research fellow and assistant scientific director in the Jewett Lab. "We set out to design a platform that could test hundreds of biosynthetic hypotheses without having to re-engineer microbes simply by mixing and matching enzymes."
Jewett likened the mix-and-match analysis of different enzyme combinations to making a cocktail.
"Imagine you're a bartender interested in making the perfect mixed drink. You would want to bring together all of the possible cocktail ingredients that potentially could be used," Jewett said. "iPROBE allows us to mix and match enzymes in this type of cocktail-based approach to determine the best combinations to carry out the transformation and synthesis of sustainable chemicals—but instead of taking months to years to do, we can do it in days to weeks."
Finding the optimal pathways in Clostridium
To validate the iPROBE system, the researchers developed optimal biosynthetic pathways for 3-hydroxybutyrate (3-HB) and butanol, two organic compounds in Clostridium autoethanogenum, a bacterium that naturally produces ethanol from metabolized carbon monoxide.
"It was important to us that we demonstrated the practical use of the technology," Karim said. "We had this dream solution to increase the pace of biotechnology research and development that could only be realized through the right collaboration."
After identifying the optimal pathways in vitro, the researchers shared them with collaborators at clean energy startup Lanzatech, which specializes in using Clostridium strains to produce sustainable fuel. Researchers there applied the pathways and found a 20-fold increase in 3-HB production in Clostridium, bridging the iPROBE's success in the lab to an industrial setting.
"Working with an organism like Clostridium is difficult; genetic tools are not as sophisticated, high-throughput workflows are often lacking, and there exist transformation idiosyncrasies," Jewett said. "To have this process work successfully pushes a new vision for sustainability. What could be better than turning waste gases from the atmosphere into sustainable chemicals at scale?"
Synthesizing limonene and styrene
In a second paper published in the journal Metabolic Engineering, Jewett and his team focused on applying iPROBE to optimize the synthesis of limonene, a member of a class of organic compounds called terpenes. Limonene is found in the oil of orange and other citrus peels and responsible for its fruity fragrance. The molecule is not only commonly used to enhance the smell of household cleaners and manufactured foods, but also has also shown the potential to help advance sustainable fuels.
In a matter of weeks, iPROBE's cell-free approach led to the exploration of hundreds of enzyme combinations to synthesize limonene.
"In the past, people have only been able to study 20 or 30 pathways," Jewett said. "We demonstrated how iPROBE could be applied to this particular biosynthetic pathway and scale not just to 100 or 200 pathways, but 500. It sets a new standard for how cell-free systems can accelerate biological design of an important sustainable chemical."
The third paper, also published in Metabolic Engineering, looked at styrene, a petroleum-derived molecule commonly used in disposable silverware and foam packaging. While past efforts have attempted to synthesize the molecule using living organisms like E. coli, styrene's natural toxicity limited production capacity. With iPROBE, Jewett and his team synthesized the highest amount of styrene through a biochemical approach to date without additional process enhancements.
"This advance opens the door to one day moving from production processes reliant on fossil fuels to more sustainable, biosynthetic-based strategies," Jewett said.
More information: Ashty S. Karim et al, In vitro prototyping and rapid optimization of biosynthetic enzymes for cell design, Nature Chemical Biology (2020). DOI: 10.1038/s41589-020-0559-0
Journal information: Nature Chemical Biology
Provided by Northwestern University