Researchers trigger enzymes with light
Enzymes are the central drivers for biochemical metabolic processes in every living cell, enabling reactions to take place efficiently. It is this ability that makes them useful as catalysts in biotechnology, for example, to create chemical products such as pharmaceutics. A topic that is currently being widely discussed is photoinduced catalysis, in which researchers harness the ability to start biochemical reactions using light. This process requires enzymes that can be activated by means of light. It is not, however, a simple matter to incorporate the few naturally occurring light-activatable enzymes into biotechnological processes, as they are highly specialised and difficult to manipulate.
Researchers at the Universities of Münster (Germany) and Pavia (Italy) have now identified an enzyme that becomes catalytically active when exposed to blue light and which immediately triggers a reaction hitherto unknown in enzymology. The reaction in question is a special monooxygenase reaction, in which an oxygen atom is transferred to the substrate. The reaction is supported by a helper molecule that delivers two electrons stepwise. Previously, it had been assumed that such a light-dependent reaction cannot occur in enzymes.
"The enzyme we have identified belongs to a very large family of enzymes, and it is realistic to assume that other enzymes that can be activated by light can be produced by means of genetic manipulation and used in a very wide range of applications," says Dr. Steffen L. Drees, who headed the study and works at the Institute of Molecular Microbiology and Biotechnology at Münster University. One possible application, for example, is the production of pharmaceuticals activated by means of light. The study has been published in the journal Nature Communications.
In their study, the researchers investigated the enzyme PqsL, which is found in the opportunistic pathogen Pseudomonas aeruginosa and, originally was not light-dependent. The researchers stimulated the enzyme with blue light and analysed the reaction using a combination of time-resolved spectroscopic and crystallographic techniques.
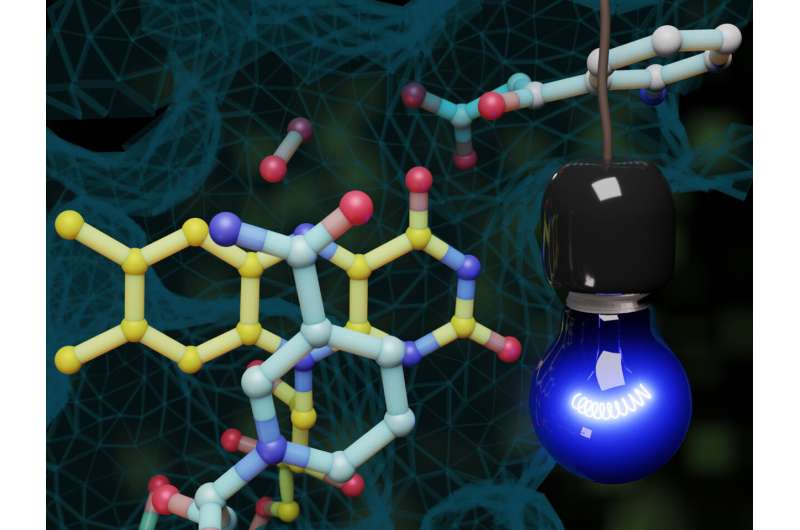
The enzyme belongs to the family of flavoproteins, and typically for this family of proteins, uses a derivative of vitamin B2 as a so-called cofactor for catalysing the incorporation of oxygen into organic molecules. The cosubstrate NADH (reduced nicotinamide adenine dinucleotide) is needed as a "helper molecule" for the enzymatic reaction, providing the necessary electrons. The reaction mechanism the researchers observed in their study was previously unknown. Activated by the exposure to light in the flavin-NADH complex, NADH transfers a single electron to the protein-bound flavin. In this way, a flavin radical is created—this is a highly reactive molecule characterised by an unpaired electron. Using time-resolved spectroscopy, the researchers were able to observe how the molecule formed and changed its state.
The flavin radical has a very negative redox potential, which means that it has a large capacity for transferring electrons to reaction partners. "Because of this property, we assume that the flavin radical can also enable additional reactions to take place, which would expand the catalytic potential of this enzyme—as well as of other enzymes too, perhaps," says group leader Prof. Susanne Fetzner.
The enzyme identified is the only one so far that is not naturally photoactive, and carries out a light-independent reaction in the bacterial cell. "The three-dimensional structure of the enzyme shows that the outward-facing flavin co-factor might be the key to photoactivation," says Simon Ernst, first author of the study.
Photoactive enzymes enable a large number of applications—for example, multi-step catalysis in a one-vessel reaction or spatially resolved catalysis, for example, to functionalise surfaces in certain patterns. They can also be useful for so-called prodrug activation in the body or on the skin—this is a process in which a pharmacological substance becomes active only after metabolization in the organism.
More information: Simon Ernst et al, Photoinduced monooxygenation involving NAD(P)H-FAD sequential single-electron transfer, Nature Communications (2020). DOI: 10.1038/s41467-020-16450-y
Journal information: Nature Communications
Provided by University of Münster