December 15, 2016 report
Method devised that allows a ketoreductase enzyme to catalyze non-natural reactions
(Phys.org)—A team of researchers working at Princeton University has come up with a new way to modify proteins to catalyze reactions that are unknown in nature. In their paper published in the journal Nature, the team outlines their new technique and the ways in which it might be useful. Uwe Bornscheuer with Greifswald University in Germany offers a News & Views piece on the work done by the team and outlines the way in which the new method is likely to impact the use of enzyme classes in bio-catalysis.
Over the years, scientists have worked to increase the number of naturally occurring enzymes that might serve as catalysts for organic synthesis because such enzymatic catalysis could, in theory, offer both high selectivity and efficiency for certain chemical reactions in intricate environments—unfortunately, only small numbers have been developed compared to the huge numbers of reaction types that are catalyzed by multiple enzymes in nature.
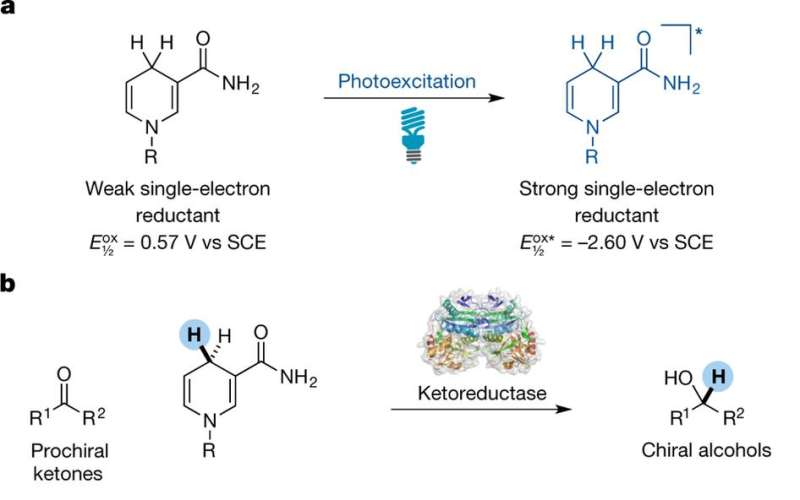
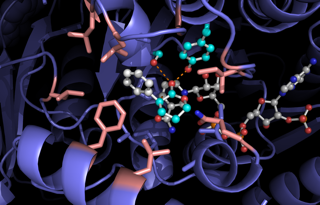
As Bornscheuer notes, thus far, researchers have come up with several ways to modify proteins to catalyze reactions that do not occur in nature, including chemical treatments and protein engineering, but more ways are needed. In this new approach, the researchers have developed a new tactic—one that works by generating enzymes using light to excite a cofactor (a molecule that increases the rate of a reaction or is required to be present for the reaction to occur) that is bound to a ketoreductase (an enzyme that catalyzes the reduction of a ketone). Once excited by the light, Bornscheuer points out, the cofactor generates a radical intermediate that can serve as a hydrogen source, one that has "handedness," which means it can be passed on to other molecules during the reaction.
As Bornscheuer also notes, because the approach devised by the team at Princeton is completely new, there exists the possibility that other types of transformations might also be effected with the introduction of light, as well. He suggests that as others begin using the new approach, they will make new findings, which means that the idea is likely to have a long-term impact on bio-catalysis and the enzymes classes that are involved.
More information: Megan A. Emmanuel et al. Accessing non-natural reactivity by irradiating nicotinamide-dependent enzymes with light, Nature (2016). DOI: 10.1038/nature20569
Abstract
Enzymes are ideal for use in asymmetric catalysis by the chemical industry, because their chemical compositions can be tailored to a specific substrate and selectivity pattern while providing efficiencies and selectivities that surpass those of classical synthetic methods. However, enzymes are limited to reactions that are found in nature and, as such, facilitate fewer types of transformation than do other forms of catalysis. Thus, a longstanding challenge in the field of biologically mediated catalysis has been to develop enzymes with new catalytic functions. Here we describe a method for achieving catalytic promiscuity that uses the photoexcited state of nicotinamide co-factors (molecules that assist enzyme-mediated catalysis). Under irradiation with visible light, the nicotinamide-dependent enzyme known as ketoreductase can be transformed from a carbonyl reductase into an initiator of radical species and a chiral source of hydrogen atoms. We demonstrate this new reactivity through a highly enantioselective radical dehalogenation of lactones—a challenging transformation for small-molecule catalysts. Mechanistic experiments support the theory that a radical species acts as an intermediate in this reaction, with NADH and NADPH (the reduced forms of nicotinamide adenine nucleotide and nicotinamide adenine dinucleotide phosphate, respectively) serving as both a photoreductant and the source of hydrogen atoms. To our knowledge, this method represents the first example of photo-induced enzyme promiscuity, and highlights the potential for accessing new reactivity from existing enzymes simply by using the excited states of common biological co-factors. This represents a departure from existing light-driven biocatalytic techniques, which are typically explored in the context of co-factor regeneration.
Journal information: Nature
© 2016 Phys.org