Researchers achieve enzyme-catalyzed Diels-Alder reaction
A computational study carried out at Zelinsky Institute of Organic Chemistry of the Russian Academy of Sciences (Moscow) focused on the mechanistic pathway of the SpnF-catalyzed cycloaddition reaction leading to Spinosyn A – tetracyclic natural insecticide produced by the cells of the bacterium Saccharopolyspora spinosa. Computational modeling revealed energetically balanced reaction coordinate of the studied process and highlighted new possibilities for enzymatic catalysis of cycloaddition reactions. The study was published in PLOS ONE journal.
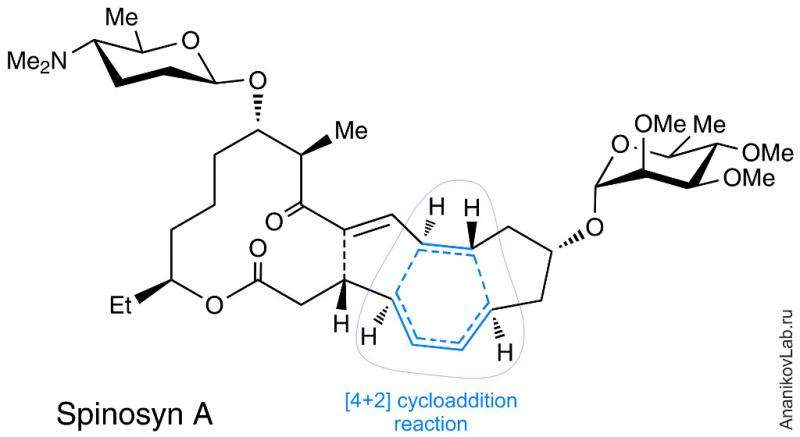
Until recently it was assumed that cycloaddition reactions (also widely known as the Diels-Alder reactions) do not occur in nature, since corresponding enzymes were never discovered. However, researchers now know that some microorganisms are capable of catalyzing challenging intramolecular [4+2] cycloaddition reaction. Living organisms have learned to apply this process: Enzyme SpnF was shown to catalyze intramolecular Diels-Alder reaction, leading to formation of natural insecticide Spinosyn A. This is a tetracyclic natural insecticide produced by the cells of the bacterium Saccharopolyspora spinosa. Indeed, [4+2] cycloaddition represents the main reaction core for assembling of polycyclic building block of Spinosyn A in this transformation (Figure 1).
When carried out in chemical laboratories, these kind of transformations are known to require harsh reaction conditions. For instance, high temperatures of around 80-120oC are required, which cannot be achieved in the living cells. Enzymatic catalysis has a unique feature – the ability to accelerate chemical reactions with the ability to carry out desired transformations at much lower temperatures. In such reactions, a catalyst is a substance that directs chemical processes of conversion of the substrate in a way that requires less activation energy for its implementation (Figure 2, left). As a result of catalysis, the reaction runs much faster than the corresponding non-catalytic process. Despite the active participation of catalysts in chemical reactions, the catalyst completely restores its structure and successfully performs acceleration again.
In the classical model of enzymatic catalysis, one stage of non-catalytic reaction with high activation energy is replaced by a multistage catalytic process with several low energy transition states and intermediate compounds (Figure 2, left). According to this well-known energy surface, it was not possible to explain catalytic cycloaddition reactions in the synthesis of Spinosyn A. Computational modeling has shown only a small decrease in activation energy, thus making a noticeable increase in reaction rate rather unlikely.
A new mechanistic pathway of the enzymatic reaction of the formation of Spinosyn A has been revealed and theoretically evaluated in the study of Prof. V.P. Ananikov and colleagues in the Zelinsky Institute of Organic Chemistry. The authors proposed a new pathway of enzyme catalysis in order to explain a greater catalytic effect.
The model assumes that the flexible cyclic molecule of the substrate may be constrained under the influence of the enzyme environment, resulting in the convergence of diene and dienophile fragments of the molecule to a certain geometry that is closer to the transition state.
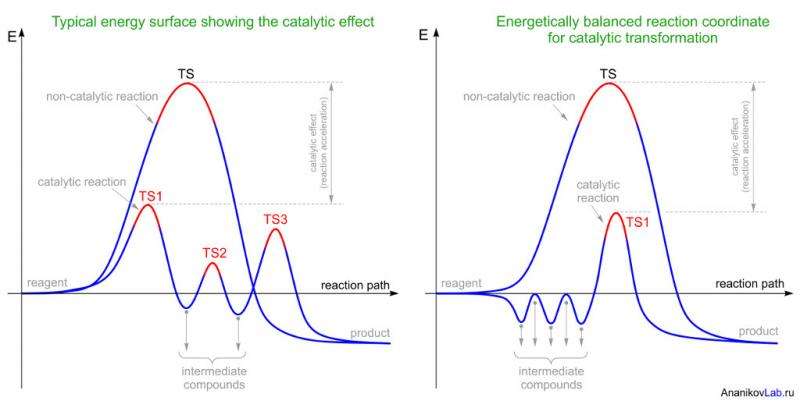
However, molecule's energy increases as a result of such compression. This increase in energy is compensated by the hydrogen bonding of the substrate with amino acid residues of the active center of the enzyme. Due to several steps of such compression/compensation, total energy of the system slightly changes, but the structure of the substrate substantially approaches the transition state. Only minor displacements of the atoms are needed in the reacting molecule in order to reach the point of transition state and turn it into the product of reaction. Thus, the overall process involves several intermediate compounds with only one transition state (Figure 2, right).
The energetically balanced reaction coordinate provides a possibility to synthesize the product by reaching a much lower activation barrier compared to a regular non-catalytic reaction pathway. It is important to note that such reaction pathway takes place only through a single transition state (Figure 2, right). These results advance our understanding of enzymatic catalysis and open new opportunities for design of new catalytic reactions.
More information: "Computational Study of a Model System of Enzyme-Mediated [4+2] Cycloaddition Reaction." PLOS ONE, 2015; DOI: 10.1371/journal.pone.0119984
Journal information: PLoS ONE
Provided by Zelinsky Institute of Organic Chemistry