May 11, 2020 feature
Measuring the charge radii of exotic copper isotopes
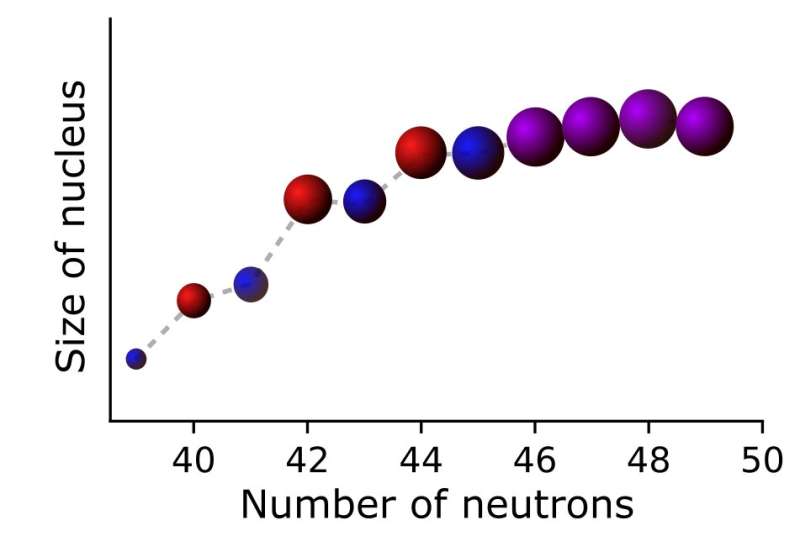
Researchers at Instituut voor Kern- en Stralingsfysica in Belgium and The University of Manchester, in collaboration with other institutes worldwide, have recently carried out a study aimed at measuring the size of the nucleus (i.e., nuclear charge radius) in neutron-rich copper isotopes. Their paper, published in Nature Physics, presents observations of a distinctive and interesting odd-even staggering pattern in the sizes of these isotopes' nuclei.
"The odd-even staggering effect we observed, where the nucleus with an odd number of neutrons is usually slightly smaller in size than their even-neutron neighbours, is more or less constant in most isotopic chains," Ruben Pieter de Groote, one of the researchers who carried out the study, told Phys.org. "In copper, however, we noticed an enhanced odd-even staggering for isotopes with approximately 40 neutrons, which then seemed to vanish when approaching 50 neutrons."
Gaining a thorough understanding of the odd-even staggering pattern observed by de Groote and his colleagues is far from an easy quest, complicated further by the fact that this pattern was found to be neutron-dependent, which was somewhat unexpected. To explore the possible reasons behind the effect they observed, the researchers performed a series of cutting-edge calculations based on nuclear theory and then compared the results of these calculations with the experimental data they collected.
"The most difficult isotope to perform measurements for, 78Cu, has 29 protons and 49 neutrons, which makes it a very complicated nucleus to study, both experimentally and computationally," de Groote said. "However, we felt that our experimental result was important enough to convince two theory collaborators to pursue two rather different theoretical methods, one based on density functionals and the other based on the valence-space in-medium similarity renormalization group method, which presents an 'ab-initio' description for medium heavy nuclei."
The two theoretical approaches that the researchers used in their study proved useful for explaining different aspects of the measurements they collected. While calculations based on density-functional theory predicted bulk properties (e.g. the total nuclear sizes) with remarkably high accuracy, the method rooted in valence-space in-medium similarity renormalization group theory provided a detailed description of the general trend for the odd-even staggering effect, as it took additional correlations into account.
"Our calculations showed that both theories contain essential ingredients for describing nuclear structure, but that there is still work to do—we still don't have a single approach that can do it all," de Groote explained.
In their recent study, the researchers focused on copper isotopes with very short lifetimes. For instance, the lifetime of 78Cu, one of the isotopes examined in their work, is of 300 milliseconds, which means that one second after it is produced the isotope will have most likely already disappeared. They thus had to use techniques that allowed them to produce and examine isotopes very quickly, before they decayed.
"What is very important when studying radioactive isotopes is that methods are fast and efficient—there is no time to collect a large sample of isotopes, to then quietly study them later on," de Groote said. "Measurements need to be performed 'on-line'; our measurement tools and detectors need to be coupled to the production site and work in perfect synchronicity."
De Groote and his colleagues used a particle accelerator known as the CERN PS-Booster, which can produce protons with a very high energy. These protons were directed to the ISOLDE facility in CERN, where they impacted onto a piece of uranium, inducing a variety of different nuclear reactions.
The nuclear reactions resulting from this process led to the production of isotopes across the entire spectrum, ranging from light helium atoms to very heavy elements, such as radium. The copper isotopes that the researchers set out to study were among these, but they had to be extracted from the large range of isotopes and purified.
"The uranium was heated up to about 2000 degrees Celsius by the ISOLDE team, so that these newly produced isotopes did not stick around, but instead escaped into an ion source: here, they were turned into charged ions," de Groote explained. "This is a crucial step, as it allows us to use electrostatic and magnetic techniques to accelerate all isotopes, select the ones of interest, and guide them to different measurement setups at the ISOLDE facility."
To measure the size of the copper isotopes, the researchers illuminated them with two separate laser beams. By tuning the frequency of the first laser in precisely the right way, they were able to to excite an electron that is bound to the nucleus. The second laser beam was then used to 'peel' this excited electron off.
"By measuring the numbers of charged particles created as we changed the frequency of the laser, we could determine the exact absorption energy of the copper atoms," de Groote said. "This absorption energy is directly related to the nuclear sizes; the shift in energy is called the isotope shift—a tiny change of colour of as low as 1 part in a million; nothing that the eye could see, but something that our system is sensitive to."
The measurement technique used by de Groote and his colleagues, known as collinear resonance ionization spectroscopy, is a highly efficient and precise tool for measuring shifts in energy in atoms. Their experimental setup is highly sophisticated, and it relies on all its different components (i.e., a large particle accelerator, ultra-stable laser systems, high-precision laser frequency measuring tools, ion traps, ultra-high vacuum pumps and high-voltage power supplies, etc.) to operate in symphony.
In their study, the researchers used it to identify 'isotope shifts' in 14 different copper isotopes. Measuring these shifts ultimately allowed them to determine changes in their size, as a function of the number of neutrons within their nucleus.
"The most challenging isotope was only produced at a rate of 20 ions per second, and in total only about 200,000 ions were used to perform the measurement," de Groote said. "The total mass of this sample, if you could collect it all before it radioactively decayed, would be 0.00000000003 microgram—compared to typical objects we are used to interacting with, this is an unbelievably small amount of stuff."
The technique employed by de Groote and his colleagues allows researchers to study isotopes that can currently only be produced in small quantities and is also far more efficient than other high-precision measurement tools developed in the past. In the future, their method could have a number of important implications for nuclear structure research, as many interesting isotopes are unstable and thus can only be produced in small quantities.
"Our results show that many of these nuclei can now be studied," de Groote says. "Further improvements of our method will push that limit even further. In particular, our technique allows us now to approach nuclei that are predicted to be produced in super- and kilonovae and have yet to be studied on earth in the laboratory. Similarly, the theoretical tools that were developed represent major milestones in nuclear theory as well."
In addition to introducing a new technique for measuring the size of nuclei in isotopes with short lifespans, de Groote and his colleagues proved the effectiveness of theoretical constructs based on density functionals and the valence-space in-medium similarity renormalization group method for studies exploring the structure of isotopes with unstable nuclei. Their study sheds some light on the advantages and disadvantages of these theoretical frameworks, which could be further explored in future studies.
"For the present study, we picked copper, since it has 29 protons," de Groote. "This makes these isotopes perfect probes to investigate the underlying nickel (28 protons) core. 78Ni (28 protons, 50 neutrons) is thought to be a doubly magic nucleus. There are only very few of these doubly-magic, closed-shell systems, and they form cornerstones for nuclear structure research, like the noble gases for atomic physics."
De Groote and his colleagues are currently working on a new study focusing on neutron-rich potassium isotopes, which have 19 protons and are thus excellent probes of magic calcium isotopes (i.e. with 20 protons). They have already performed initial calculations of the charge radii of these isotopes and now plan to investigate these results more in depth.
"On the longer term, a measurement campaign on indium and tin isotopes, close to the doubly-magic 100Sn and 132Sn isotopes, was already initiated, and will be pursued in the coming years," de Groote says. "These isotopes are on the current frontier for nuclear theories; experimental and theoretical efforts are thus progressing nicely in tandem."
De Groote and his colleagues have also started using the same experimental method introduced in their recent paper to study radioactive molecules. For instance, they recently completed the very first spectroscopic study of radium-fluoride, a molecule that contains a radioactive radium atom.
"Since there are no stable radium isotopes, this molecule could never be studied before," de Groote explained. "This is particularly exciting, as it may be the key to the next generation of research for physics beyond the standard model."
More information: R. P. de Groote et al. Measurement and microscopic description of odd–even staggering of charge radii of exotic copper isotopes, Nature Physics (2020). DOI: 10.1038/s41567-020-0868-y
Journal information: Nature Physics
© 2020 Science X Network





















