Transposable elements play an important role in genetic expression and evolution
Until recently, little was known about how transposable elements contribute to gene regulation. These are little pieces of DNA that can replicate themselves and spread out in the genome. Although they make up nearly half of the human genome, these were often ignored and commonly thought of as "useless junk," with a minimal role, if any at all, in the activity of a cell. A new study by Adam Diehl, Ningxin Ouyang, and Alan Boyle, University of Michigan Medical School and members of the U-M Center for RNA Biomedicine, shows that transposable elements play an important role in regulating genetic expression with implications to advance the understanding of genetic evolution.
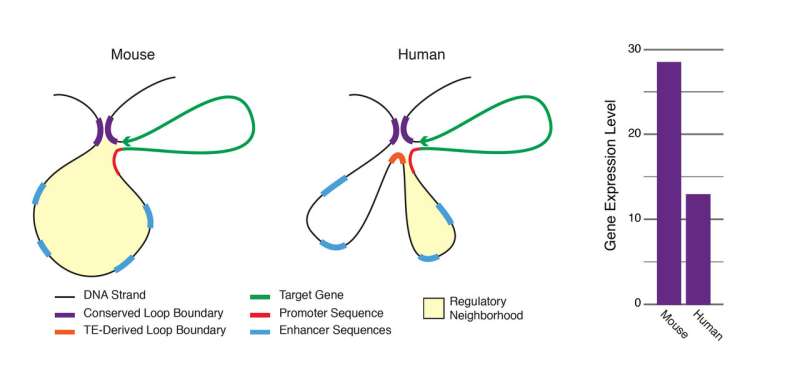
Transposable elements move around the cell, and, unlike previously thought, the authors of this paper found that when they go to different sites, transposable elements sometimes change the way DNA strands interact in 3-D space, and therefore the structure of the 3-D genome. It appears a third of the 3-D contacts in the genome actually originate from transposable elements leading to an outsized contribution by these regions to looping variation and demonstrating their very significant role in genetic expression and evolution.
The main component that determines 3-D structure is a protein called CTCF. This study particularly focused on how transposable elements create new CTCF sites that, in turn, hijack existing genomic structure to form new 3-D contacts in the genome. The authors show that these often create variable loops that can influence regulatory activity and gene expression in the cell. These findings were observed in human cells and mouse cells and show how transposable elements contribute to intraspecies variation and interspecies divergence, and will guide further research efforts in areas including gene regulation, regulatory evolution, looping divergence, and transposable element biology.
To streamline this work, the authors developed a piece of software, MapGL, to track the physical gain and loss of short genetic sequences across species. For example, a sequence that existed in the most common ancestor may have been lost somewhere or, inversely, could have been absent in the common ancestor but later gained in the human genome. MapGL enables predictions about the evolutionary influences of structural variations between species and makes this type of analysis much more accessible. For this paper, their input was a set of CTCF binding sites which were labeled by MapGL to show that a sequence gain/loss process explains many of the differences in CTCF binding between humans and mice.
With a background in computer science and molecular biology, Alan Boyle explains that he has always been interested in gene regulation. "It's like a complex circuit: perturbing gene regulation through changes to the three-dimensional structure of the genome can have very different and wide-ranging outcomes."
For Adam Diehl, this research continues the great discoveries that started in the late 1800s, when scientists first looked at the shape of chromosomes through microscopes. They observed the shape differences between cells, and noticed that the shape inside the nuclei remained the same between mother and daughter cells. Decades later, transposable elements were discovered at his alma mater, Cornell University: jumping genes could change the phenotypes of corn plants. In the 70s, because the genes between humans and chimpanzees are much too similar to explain the differences between the species, scientific focus shifted on how the genes are being used. For Diehl "It's so exciting to be able to synthesize all this knowledge, and contribute to the next step of the story of species evolution."
This research team will further study the impact of transposable elements on the 3-D genome, but this time with a particular interest on a single human population sample rather than across species. The next steps will include experimental follow-up using a new sequencing method capable of identifying transposable element insertions that are variable across human populations. This method was developed in collaboration with Ryan Mills's lab, at the University of Michigan, Medical School. It is expected that the next results will further the understanding of the regulatory role of the transposable elements with possible applications to neurodegenerative diseases.
More information: Adam G. Diehl et al, Transposable elements contribute to cell and species-specific chromatin looping and gene regulation in mammalian genomes, Nature Communications (2020). DOI: 10.1038/s41467-020-15520-5
Journal information: Nature Communications
Provided by University of Michigan