COVID-19 puts new science to the pressure test
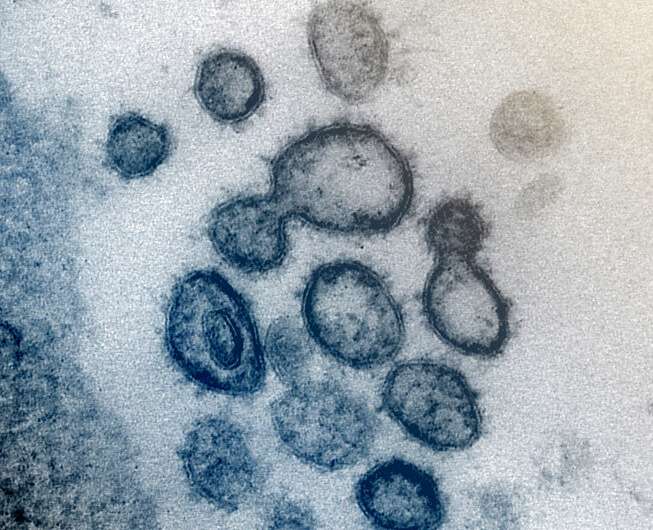
By its very nature, science rarely offers a quick fix. New technologies and medicines often take years to prove that they are safe and effective. Yet the surging COVID-19 pandemic is forcing scientists to condense this process to mere months. Researchers are working at breakneck speeds to meet the challenge, according to a series of articles in Chemical & Engineering News (C&EN), the weekly newsmagazine of the American Chemical Society.
While many scientists are testing existing drugs, others are working to develop brand-new diagnostics, treatments and vaccines fine-tuned to fight the novel coronavirus. Many of these strategies involve relatively new technologies, like gene-based vaccines or the gene-editing tool CRISPR, according to Senior Correspondent Lisa Jarvis. Because of the urgency to get new tests, therapies and preventions into the clinic amid a pandemic, many of these approaches are undergoing a trial by fire, Jarvis writes.
Experts agree that widespread testing is key to flattening the curve of the COVID-19 pandemic, but the U.S. and other countries have experienced diagnostic bottlenecks. Therefore, some researchers are working to develop new tests that can be conducted more quickly and easily than existing tests based on the polymerase chain reaction, or PCR. In terms of therapies, several clinical trials for antibody-based treatments, such as convalescent plasma, hyperimmune therapy and monoclonal antibodies, for COVID-19 are set to begin within the next few months. And on the prevention front, new types of DNA and mRNA vaccines—which can be designed much more quickly than traditional vaccines—are now set for clinical trials. According to leading scientists, the coronavirus pandemic will likely transform the pace and priorities of science for years to come.
More information: "Coronavirus puts science to the test," cen.acs.org/biological-chemist … -science-test/98/i14
Provided by American Chemical Society





















