Opening a new chapter in antibody mimetics
Antibodies are proteins that act as recognition molecules for pathogens, like viruses and bacteria, and are the workhorses of the body's immune system. They recognize specific molecules and bind to them very strongly, which makes them ideal for biomedical applications like diagnostics or therapeutic treatments. Unfortunately, production of antibodies is expensive, and they aren't very stable. This has motivated a growing number of chemists to explore new synthetic materials that can mimic key aspects of antibody structure and function. Nonetheless, it remains a fundamental challenge to create chemically diverse populations of protein-like, folded synthetic nanostructures that can be tailored to specifically bind pathogens and other molecules.
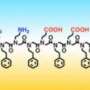
In a recent study published in ACS Nano, a team of Foundry staff, working closely with users from UC San Francisco, Pacific Northwest National Laboratory, and New York University, have developed a new method to rapidly synthesize and screen libraries of two-dimensional peptoid nanostructures capable of selectively binding target proteins. Peptoids are bioinspired, sequence-defined molecules that act as building blocks for constructing protein-like structures.
"We can now readily build populations of synthetic materials that can be engineered to recognize a potential pathogen," said Ron Zuckermann, a Senior Scientist at the Foundry who led the study. "It is a shining example of biomimetic nanoscience: creating functional chemical architectures from folded, information-rich polymer chains."
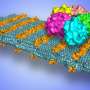
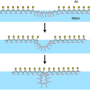
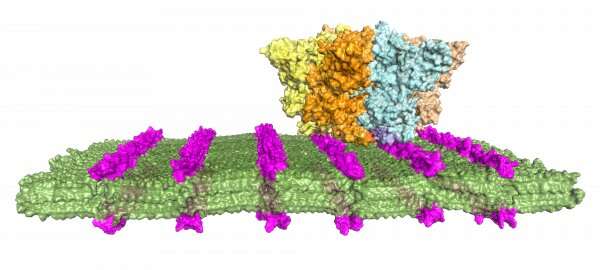
Zuckermann and his colleagues designed a family of peptoid polymers to fold into ordered nanosheets displaying a high density of diverse peptoid loops on their surface, like a molecular version of Velcro. The density of loops on the nanosheet offers multiple sites of attachment to target proteins and boosts binding selectivity and sensitivity.
One of the major limitations to screen large libraries of folded nanostructures is the complexity of their synthesis. The research team worked to automate nearly every step of the synthesis and screening process—from the chemical synthesis of loop-containing peptoids (loopoids), assembling the loopoids into nanosheets, screening the loopoid nanosheets against various proteins for binding activity (hits), and validating hits.
Using this new procedure, the researchers identified a peptoid structure that readily and selectively binds to anthrax protective antigen, a toxin-related protein.
"This work was the result of a huge effort from a number of institutions and represents a milestone for the field," said Zuckermann.
Optimization of the synthesis, assembly, and screening processes provides a scalable strategy for generating and screening large chemical libraries of 2-D nanomaterials that can exhibit potent and selective binding to target proteins. These properties should allow for the rapid discovery of pathogen-specific binding materials and have an impact on many biomedical applications such as sensing, diagnostics, and therapeutics.
More information: Jae Hong Kim et al. Discovery of Stable and Selective Antibody Mimetics from Combinatorial Libraries of Polyvalent, Loop-Functionalized Peptoid Nanosheets, ACS Nano (2019). DOI: 10.1021/acsnano.9b07498
Journal information: ACS Nano
Provided by Lawrence Berkeley National Laboratory