Monitoring organs and cells in living fly larvae
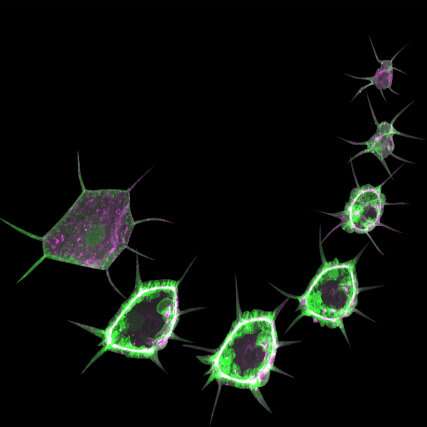
Small changes in a cell's composition can radically transform its function and drive the development of diseases like diabetes, cancer or neuronal dysfunction. Scientists led by the European Molecular Biology Laboratory in Heidelberg, the Institute for Genetics of the University of Cologne, the Max Planck Institute for Biology of Ageing, and CECAD and the CMMC in Cologne have developed a method to study processes taking place in the cells of a living fruit fly larva. This technique, published in Nature Protocols on 10 February, provides a simple but effective way to study the functions of organs in living animals.
For decades, scientists have been using the fruit fly Drosophila to understand biological processes. Key biological processes and 60% of fruit fly genes are also seen in humans, having been evolutionarily conserved. The fruit fly is therefore a powerful model organism for studying genetics. Scientists have created many genetic tools and techniques to study molecular processes of development and human disease in the fruit fly.
The challenge
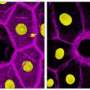
Most previous studies have been carried out in the Drosophila embryo or in adult flies, but the fruit fly larva also offers enormous research potential. Its transparent body, with highly developed functional organs including brain, gut, and muscle, makes the larva an excellent system for observing the dynamics of cellular and molecular processes in living animals.
"The cells in a fruit fly larva are much bigger than in the embryo. You can see all organelles and even the subcellular processes. That's the beauty of the system," says Parisa Kakanj, the scientist who led the study in Germany.
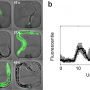
However, the larva's continuous crawling makes live monitoring a challenge, and some cellular processes only reveal their secrets through long-term imaging. Kakanj, a researcher in the groups of Maria Leptin and Linda Partridge, tackled this challenge and developed a method for simple immobilization of the larvae that allows long-term live imaging and the investigation of cellular events at high resolution.
Simple, fast, and efficient
In the past, scientists have used mechanical methods or anesthetics to tranquilize fruit fly larvae. Both approaches had undesired side-effects, such as mechanical pressure, limited immobilization, and high mortality. Further, simultaneous screening of many larvae was impossible.
"In contrast to previous techniques, we developed a simple method for short-term treatment with ether, a classical anesthetic. This approach allowed undisrupted long-term immobilization of many larvae in parallel," explains Parisa Kakanj.
A promising model for drug discovery
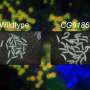
To prove the technique's capabilities, the team investigated wound healing in larvae. They examined the influence of insulin and TOR—crucial signaling molecules for cell survival, growth, and proliferation—on this process. By studying the subcellular dynamics, the scientists created a temporal map of insulin and TOR pathway activity during wound closure. They found that lowering insulin signaling at the wound edge slows down the healing process.
Long-term in vivo imaging, coupled with genetic manipulations, opens the path to many aspects of biology and physiology. "This technique will help many scientists studying neuronal signaling, fat metabolism, or tumor formation, and will provide new opportunities for drug discovery," says Parisa Kakanj.
More information: Parisa Kakanj et al. Long-term in vivo imaging of Drosophila larvae, Nature Protocols (2020). DOI: 10.1038/s41596-019-0282-z
Journal information: Nature Protocols
Provided by European Molecular Biology Laboratory