November 22, 2019 report
Using a two-step approach to convert aliphatic amines into unnatural amino acids
A team of chemists at Nankai University has developed a two-step approach to converting aliphatic amines into unnatural amino acids. In their paper published in the journal Science, the group describes their approach, how well it worked, and applications that might benefit from its use. John Ovian and Eric Jacobsen with Harvard University have published a companion piece in the same journal issue outlining some of the obstacles to forging bonds with rich nitrogen reactants; it also describes the approach used by the team in China.
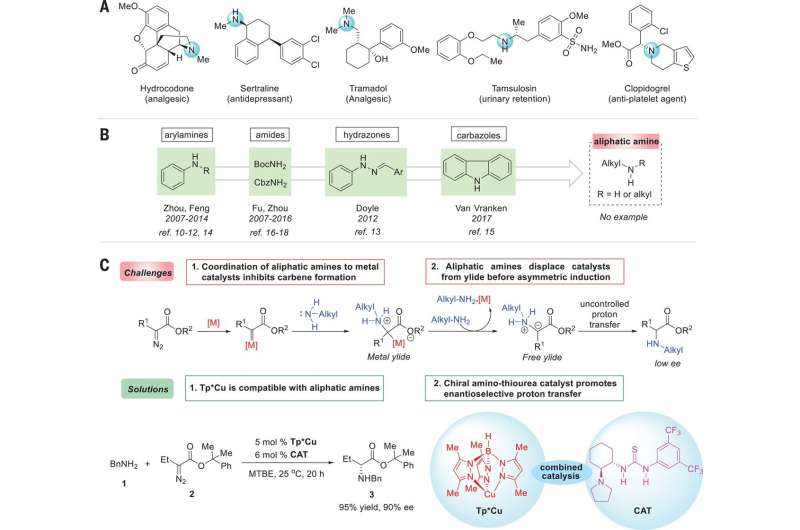
The researchers began by noting that chiral amines are used in a wide variety of natural products, and point out that they are also just as widely used in agrochemicals and pharmaceuticals. They note that 43 percent of the 200 most prescribed prescription medications in 2016 had at least one analiphatic amine moiety. They further note that because of this, developing enantioselective transition-metal-catalyzed reactions that form C–N bonds are of great interest in chemistry circles.
As Ovian and Jacobsen note, chemists use a variety of compounds that have carbon-nitrogen bonds in one of two possible mirror-image orientations. But as they also point out, doing so can present difficulties due to the nitrogen interacting with a given catalyst. In this new effort, the researchers have found a way around this problem by developing a two-step approach to converting aliphatic amines into unnatural amino acids. In their approach, they applied a copper catalyst to serve as a means for activating a carbon reactant—then added a thiourea catalyst that was hydrogen bonding to produce high-selectivity products. They note that the success of the transformation was reliant on the unique properties of both catalysts. They further report that the reaction that resulted should work equally well with a wide range of diazo derivatives of an ester/amine coupling partners.
Ovian and Jacobsen further note that pairing an amine with a carbenoid allowed for stabilizing by a carboxyl group and provided a good way to create unnatural α–amino acid derivatives. The method also plots a path for extending metal-catalyzed carbenoid insertions into N-H bonds to aliphatic amines and represents the potential of developing new asymmetric transformations.
More information: "Highly enantioselective carbene insertion into N–H bonds of aliphatic amines" Science (2019). science.sciencemag.org/cgi/doi … 1126/science.aaw9939
Journal information: Science
© 2019 Science X Network