August 21, 2019 feature
Self-assembled membrane with water-continuous transport pathways for precise nanofiltration
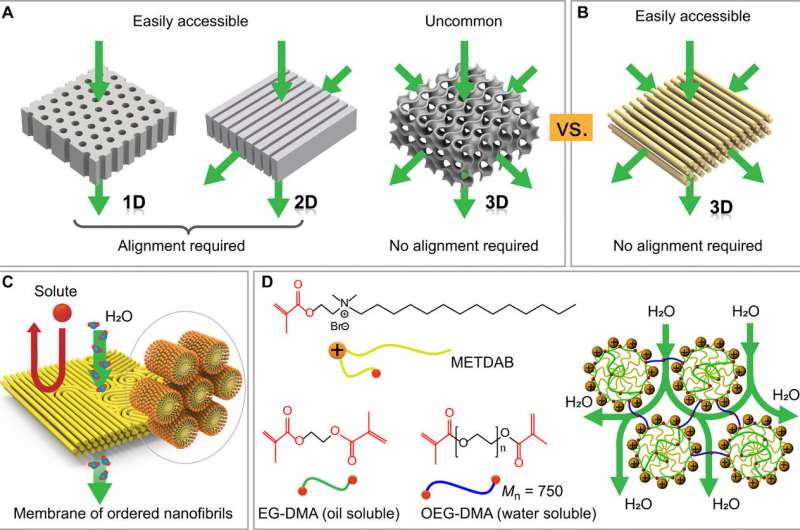
Self-assembled materials are attractive for next-generation materials, but their potential to assemble at the nanoscale and form nanostructures (cylinders, lamellae etc.) remains challenging. In a recent report, Xundu Feng and colleagues at the interdisciplinary departments of chemical and environmental engineering, biomolecular engineering, chemistry and the center for advanced low-dimension materials in the U.S., France, Japan and China, proposed and demonstrated a new approach to prevent the existing challenges. In the study, they explored size-selective transport in the water-continuous medium of a nanostructured polymer template formed using a self-assembled lyotropic H1 (hexagonal cylindrical shaped) mesophase (a state of matter between liquid and solid). They optimized the mesophase composition to facilitate high-fidelity retention of the H1 structure on photoinduced crosslinking.
The resulting nanostructured polymer material was mechanically robust with internally and externally crosslinked nanofibrils surrounded by a continuous aqueous medium. The research team fabricated a membrane with size selectivity at the 1 to 2 nm length scale and water permeabilities of ~10 liters m−2 hour−1 bar−1 μm. The membranes displayed excellent anti-microbial properties for practical use. The results are now published on Science Advances and represent a breakthrough for the potential use of self-assembled membrane-based nanofiltration in practical applications of water purification.
Membrane separation for filtration is widely used in diverse technical applications, including seawater desalination, gas separation, food processing, fuel cells and the emerging fields of sustainable power generation and distillation. During nanofiltration, dissolved or suspended solutes ranging from 1 to 10 nm in size can be removed. New nanofiltration membranes are of particular interest for low-cost treatment of wastewaters to remove organic contaminants including pesticides and metabolites of pharmaceutical drugs. State-of-the-art membranes presently suffer from a trade-off between permeability and selectivity where increased permeability can result in decreased selectivity and vice-versa. Since the trade-off originated from the intrinsic structural limits of conventional membranes, materials scientists have incorporated self-assembled materials as an attractive solution to realize highly selective separation without compromising permeability.
For example, block copolymers (BCPs) and small-molecule liquid crystals (LCs) can self-assemble into a series of mesophase forms with periodic nanoscale domains to contain thermodynamically defined sizes and shapes. The well-ordered nanostructures found in BCPs and LCs in the form of cylinders, lamellae and gyroids are attractive templates to fabricate nanopore membranes. Self-assembling materials could provide useful templates to control the organization of discrete objects such as water channel proteins or nanotubes, to function as nanofiltration pores. Although the nanopore membranes showed high selectivity and permeability for nanofiltration and ultrafiltration, scientists have encountered challenges during their practical development.
In the present work, Feng et al. developed a scalable approach to obtain highly permeable and selective nanofiltration membranes with attractive anti-biofouling properties for antimicrobial activities. The membrane contained hexagonally ordered molecular nanofibrils realized by crosslinking a direct cylindrical lyotropic LC. The membrane was mechanically robust and resilient against both dehydration and swelling by excess water. The self-assembled structure provided uniform and well-defined spacing between nanofibrils for high membrane selectivity and reduced membrane complexity, departing from previously reported work. The new characteristics set the membranes apart from nanostructured membranes derived from lyotropic LCs reported to date, to offer a path toward practical nanofiltration.
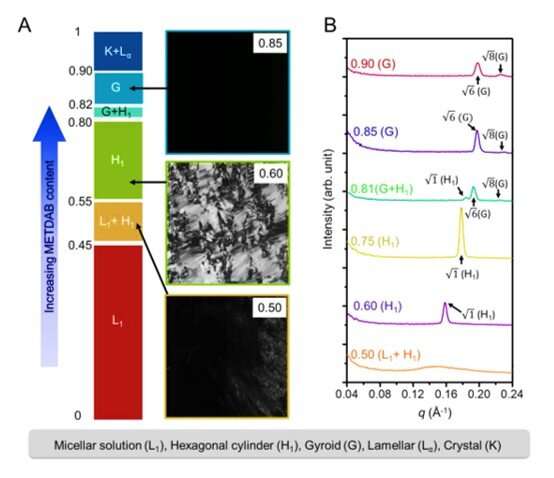
To form a polymerizable H1 mesophase with water, the research team used a cationic surfactant, 2-(methacryloyloxy)ethyl tetradecyl dimethyl ammonium bromide (METDAB) alongside additional crosslinkers of ethylene glycol dimethacrylate (EG-DMA) and oligo(ethylene glycol) dimethacrylate (OEG-DMA). The research team synthesized the surfactant monomer or surfmer in a single-step Menshutkin reaction and formed H1 phases at room temperature using METDAB concentrations ranging from 55 to 80 weight percentage.
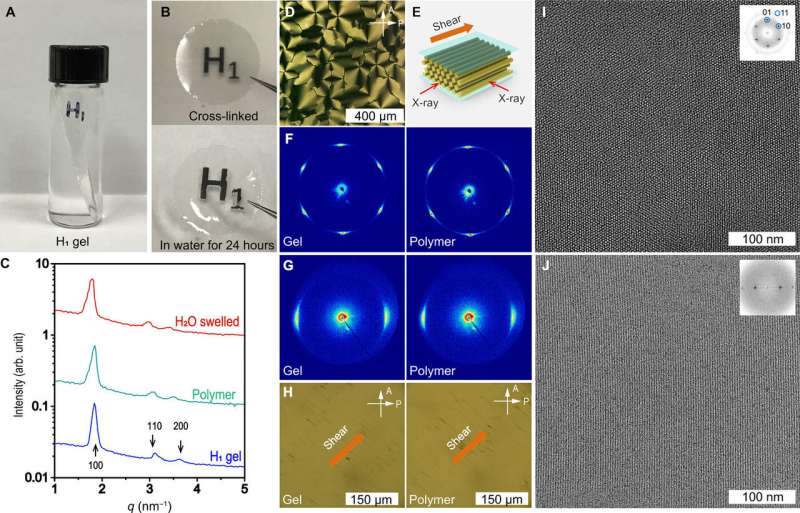
They performed detailed structural characterizations using high-resolution small-angle X-ray scattering (SAXS) with high-resolution microscopy to verify the retention of the H1 mesophase after UV-initiated cross-linking. They optimized the composition to develop 70 weight percent METDAB with 22. 8 weight percent water, 5.4 weight percent OEG-DMA and 1.8 weight percent EG-DMA with photoinitiator to form a stable, homogenous H1 mesophase gel. The resulting gel sample showed excellent transparency to retain integrity after immersing in water for 24 hours for resistance against structural collapse due to water swelling. Feng et al. obtained a high-resolution TEM image of the sample to observe an ordered array of hexagonally packed nanofibrils displaying a six-fold symmetry.
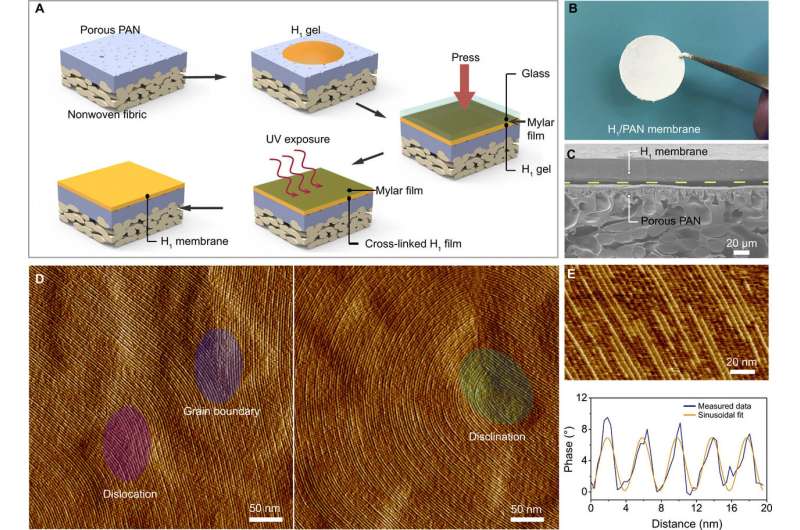
The water-continuous nature of the self-assembled, cross-linked H1 mesophase and its mechanical resilience was attractive for membrane application, which Feng et al. tested as a proof-of-concept. For this, they produced a membrane by cross-linking a thin film of the H1 mesophase with UV exposure, on a commercially sourced, polyacrylonitrile (PAN) ultrafiltration membrane. They then imaged the H1/PAN composite membrane using a scanning electron microscope (SEM) and tested the surface morphology using atomic force microscopy (AFM). During the experiments, the composite membranes showed strong size selectivity toward charged organic dye molecules. Based on the transport data, Feng et al. showed that the membrane could effectively separate solutes on the basis of their size and charge.
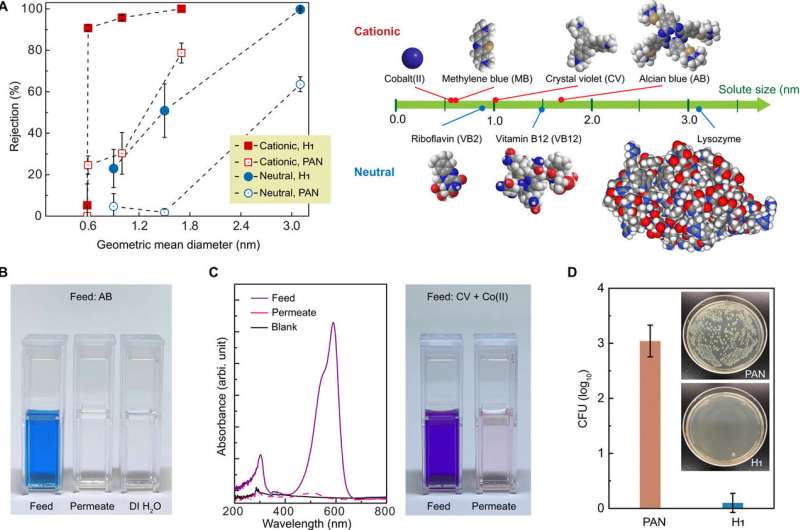
The researchers propose additional investigations to highlight the boundary between porous and solution diffusion mechanisms in polymer membranes. For instance, the water permeability decreased up to 50 percent when they filtered charged solutes but remained unaltered in the presence of natural solutes. The permeabilities and function of the self-assembled nanomembranes developed in the study compared favorably with commercial nanofiltration membranes such as Dow FILMTEC NF90-400. The research team observed water-facing quaternary ammonium groups on the nanofibrils to represent anti-biofouling behavior due to well-established anti-microbial properties of the functional groups. They studied the potential for anti-biofouling behavior of H1 membranes using a standard colony-forming unit (CFU) enumeration assay.
During the experiments, they kept PAN membranes (control group) or H1 membranes in contact with gram-negative bacteria (Escherichia coli) in suspension for 3 hours. When they sonicated the membranes mildly in saline solution, the bacteria detached from the surfaces for subsequent culture on agar and overnight incubation. The results showed the number of viable E. coli cells on the H1 membrane to be 3 orders of magnitude smaller than the control, proving anti-biofouling activity of the membranes. The results confirmed a strong antimicrobial response for H1, as anticipated due to the presence of quaternary ammonium groups.
In this way, Xundu Feng and colleagues reported a simple approach to engineer polymer nanofiltration membranes with a unique structure of ordered nanofibril arrays. In the approach, they used a cross-linkable, water-continuous lyotropic H1 mesophase template to create the morphology of interest. They followed the engineering step with systematic, materials characterization techniques to confirm the formation of highly ordered nanostructures with high fidelity for mechanical robustness. The present work was the first to directly image the polymerized lyotropic H1 mesophase.
The scientists synthesized the main constituent species of the system METDAB in a single step, using readily available and inexpensive reagents. Using the membrane, they demonstrated clear, size-based selectivity with molecular dyes as model solutes, alongside water permeabilities approximating 10 liters m−2 hour−1 bar−1 μm. The research team aims to optimize the procedures of fabrication to provide thinner selective layers by modifying the surface chemistry of the nanofibrils for water nanofiltration and treatment using antimicrobial membranes in practical applications.
More information: Xunda Feng et al. Precise nanofiltration in a fouling-resistant self-assembled membrane with water-continuous transport pathways, Science Advances (2019). DOI: 10.1126/sciadv.aav9308
Ho Bum Park et al. Maximizing the right stuff: The trade-off between membrane permeability and selectivity, Science (2017). DOI: 10.1126/science.aab0530
D. L. Gin et al. Designing the Next Generation of Chemical Separation Membranes, Science (2011). DOI: 10.1126/science.1203771
Bruce E. Logan et al. Membrane-based processes for sustainable power generation using water, Nature (2012). DOI: 10.1038/nature11477
Journal information: Science Advances , Science , Nature
© 2019 Science X Network





















