Scientists advance search for memory's molecular roots
A new piece of a difficult puzzle—the nature of memory—fell into place this week with a hint at how brain cells change structure when they learn something.
Interactions between three moving parts—a binding protein, a structural protein and calcium—are part of the process by which electrical signals enter neural cells and remodel the molecular structures thought to enable cognition and the storage of memories.
Colleagues from Rice University, the University of Houston (UH) and The University of Texas Health Science Center at Houston (UTHealth) combined theories, simulations and experiments to determine how a central binding protein—calcium-calmodulin-dependent kinase II (CaMKII)—binds and unbinds from the cytoskeleton of a neuron.
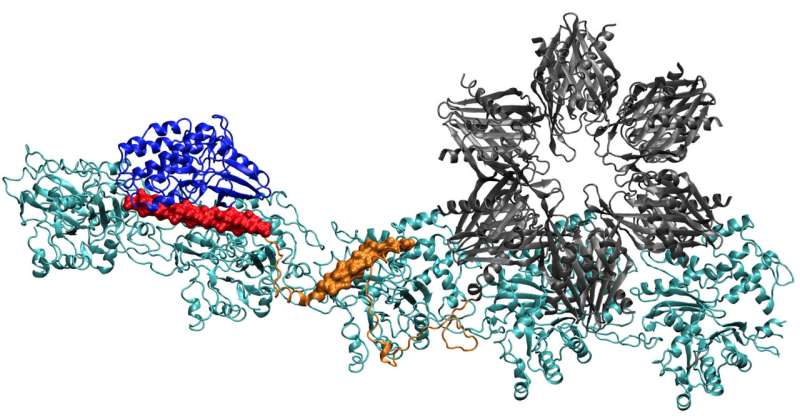
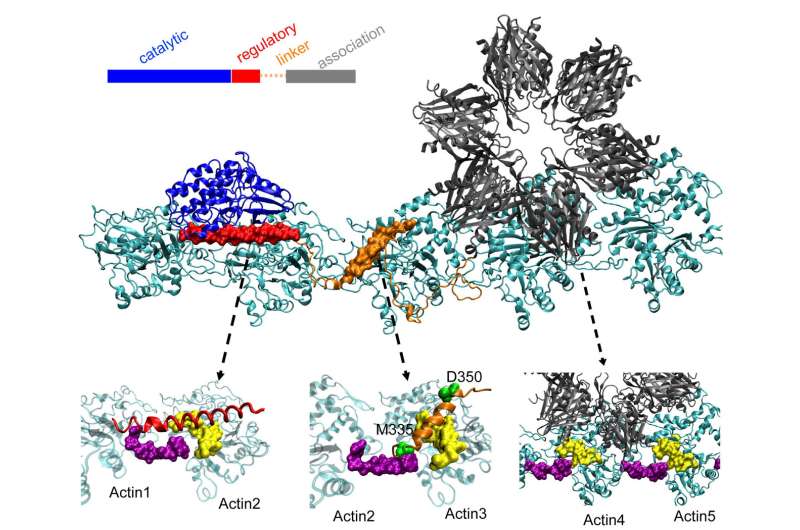
The team's report in the Proceedings of the National Academy of Sciences gives the first clear details of how the binding sites of CaMKII act to align actin filaments—the structural protein—into long, rigid bundles. The bundles serve as the supporting skeletons of dendritic spines, spiky protrusions that receive chemical messages through synapses from other neurons.
Peter Wolynes, a theoretical physicist at Rice, joined an ongoing collaboration by UH physicist Margaret Cheung and UTHealth neurobiologist Neal Waxham that aimed to understand how signals make their way through dendrites, the branches on nerve cells that transmit information between cells.
Finding the complete structure of CaMKII has proven too complex for X-ray crystallography, although parts of its structure were known. When combined with the actin that makes up the cytoskeleton, the system also became the largest protein Wolynes and his team have analyzed via their protein-structure prediction program, AWSEM.
When they were done, the structure predicted by the computer was a remarkable match for two-dimensional electron microscope images by Waxham and his group that clearly show parallel actin filaments are held together, ladder-like, by rungs of CaMKII.
"There definitely are preliminary chemical steps involving the enzyme activity of CaMKII before you get to this stage; therefore, we don't have a completely clear picture of how to put everything together," Wolynes said. "But it's clear the assembly of the complex is the key step where chemistry turns into a larger-scale structure that can hold a memory."
CaMKII is uniquely suited to interact with actin, the most abundant protein in eukaryotic cells and one that has special abilities in neurons, where it not only has to give thousands of dendrites (in each of billions of neurons) their resting forms but also must give them a level of plasticity to adapt to a constant barrage of signals.
Actin molecules self-assemble into long, twisting filaments. The hydrophobic pockets between these molecules are perfectly configured to bind CaMKII, a large protein with multiple parts, or domains. These domains lock in to three consecutive binding sites on the filament, and the twists put binding sites at regular intervals to keep the proteins from piling up.
CaMKII's "association" domain is a six-fold subunit that also binds to adjacent filaments to form actin bundles, the backbones of dendritic spines that give these protrusions their shapes.
These bundles remain rigid if the dendrite contains little calcium. But when calcium ions enter through the synapse, they combine with calmodulin proteins, allowing them to bind to another part of CaMKII, the floppy regulatory domain. That triggers the disassociation of a domain of CaMKII from the filament, followed by the rest of the protein, opening a short window of time during which the bundles can reconfigure.
"When enough calcium comes in, the activated calmodulin breaks up these structures, but only for a while," Wolynes said. "Then the cytoskeleton reforms. During that time, the dendritic spine can take on a different shape that might be bigger."
"We know calcium brings information into the cell," Cheung added. "But how nerve cells know what to do with it really depends on how this protein encodes information. One part of our work is to connect that on a molecular level and then project how these simple geometrical rules develop larger microscale structures."

The team's calculations showed the association domain is responsible for about 40% of the protein's binding strength to actin. A linker domain adds another 40% and the crucial regulatory domain provides the final 20%—a sensible strategy, since the regulatory domain is on the lookout for incoming calcium-calmodulins that can unzip the entire protein from the filament.
The project came together through Rice's Center for Theoretical Biological Physics (CTBP), of which Wolynes is co-director and Cheung a senior scientist. Their association goes back to when both were at the University of California, San Diego, he as a professor and she as a graduate student of Rice physicist José Onuchic, also a CTBP co-director. Wolynes also served on her thesis review panel, she said.
Cheung was aware of previous work by Wolynes and his Rice group that suggested actin stabilizes prion-like fibers thought to encode memories in neurons and decided it was a good match for her research with Waxham to see how calcium activates CaMKII.
"This is one of the most interesting problems in neuroscience: How do short-term chemical changes lead to something long term, like memory?" Waxham said. "I think one of the most interesting contributions we make is to capture how the system takes changes that happen in milliseconds to seconds and builds something that can outlive the initial signal."
The puzzle is far from complete, Wolynes said. "The earlier work by Margaret and Neal was about the initiation of memory events," he said of his colleagues' study of calmodulin. "Our prion paper was on the preservation of memory, at the end of the learning process. And actin's in the middle. There may be many other things in the middle, too.
"These big-picture questions are interesting to a lot of people," he said. "This is a key element of the problem, but it's clearly not the end of the story."
More information: Qian Wang el al., "Calcium/calmodulin-dependent kinase II – Actin assemblies and their dynamic regulation by calmodulin in dendritic spines," PNAS (2019). www.pnas.org/cgi/doi/10.1073/pnas.1911452116
Journal information: Proceedings of the National Academy of Sciences
Provided by Rice University