A new path to cancer therapy: Developing simultaneous multiplexed gene editing technology
Dr. Mihue Jang and her group at the Center for Theragnosis of the Korea Institute of Science and Technology (KIST, President Byung-gwon Lee) announced that they have developed a new gene editing system that could be used for anticancer immunotherapy through the simultaneous suppression of proteins that interfere with the immune system expressed on the surface of lymphoma cells and activation of cytotoxic T lymphocyte, based on the results of joint research conducted with Prof. Seokmann Hong and his group at Sejong University (President, Deg-hyo Bae).
Gene editing technology is a technology that eliminates the underlying causes of and treats diseases by removing specific genes or editing genes to restore their normal function. In particular, CRISPR gene editing technology is now commonly used for immunotherapy by correcting the genes of immune cells to induce them to attack cancer cells selectively.
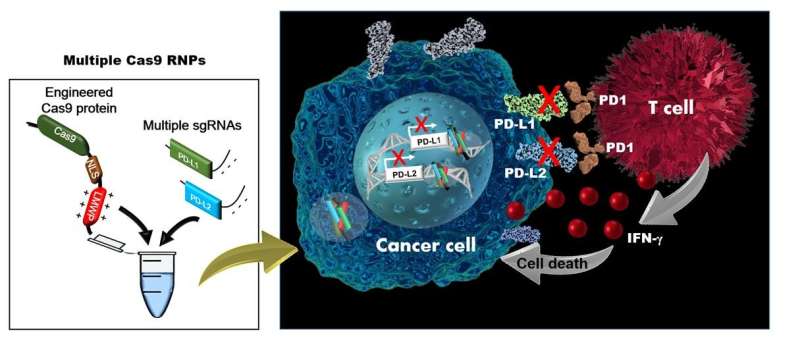
Dr. Mihue Jang of KIST improved the CRISPR gene editing system to enable the penetration of the cell membrane without external carriers (ACS Nano 2018, 12, 8, 7750-7760). However, there are various kinds of genes that regulate immune activity, and technology for inducing safe and convenient immunotherapy is not yet sufficiently developed. The collaborative research team of Dr. Jang's group at KIST and Prof. Hong' group at Sejong University developed a technology that is applicable to immunotherapy by further improving the CRISPR gene editing system to allow for the transfer of genes to lymphoma cells without external carriers as well as the correction of several genes at the same time.
Existing gene editing technology has been used to transfer genes into lymphoma cells, such as T lymphocytes, among immune cells, mainly using the viral transduction or electroporation methods. The viral transduction method often induces undesired immune responses and has a high chance of inserting genes into the wrong genome sequences. Also, the electric shock method requires separate and expensive equipment, has great difficulty correcting large numbers of cells at one time, and shows low cell viability.
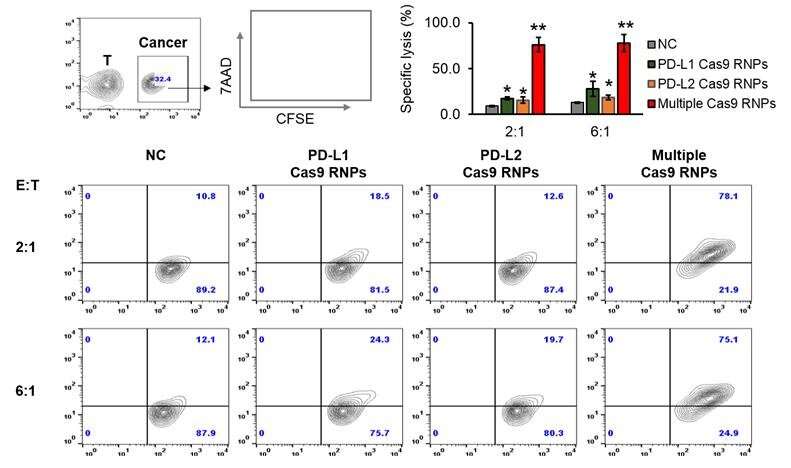
This technology jointly developed by research teams of KIST and Sejong University simultaneously targets PD-L1 and PD-L2, among the inhibitory immune checkpoints known for suppressing the immune system. The treatment efficacy was confirmed, showing that targeting these immune checkpoints does not interfere with the immune system and that the cytotoxic T lymphocytes directly attack cancer cells to increase the anticancer immune response.
Dr. Mihue Jang of KIST said, "This newly developed gene editing system can be applied to various types of immune cells, and is thus expected to be used in the development of treatments for various diseases, including not only cancers but also autoimmune and inflammatory diseases."
More information: Anna Ju et al, A carrier-free multiplexed gene editing system applicable for suspension cells, Biomaterials (2019). DOI: 10.1016/j.biomaterials.2019.119298
Journal information: ACS Nano , Biomaterials
Provided by National Research Council of Science & Technology