Novel reusable hydrogel could harvest clean, desalinated water from humid air above sea surfaces

Huge amounts of water evaporate from water surfaces such as reservoirs and the open sea each day. To address this issue, especially in water scarce areas, researchers from the National University of Singapore (NUS) found an innovative way to harvest water from the humid air above water surfaces.
The solution lies in a unique zinc-based water-absorbing hydrogel that was developed last year by a research team at the NUS Faculty of Engineering. This gel-like material is over eight times more absorbent than commercial drying agents and only natural sunlight is required to harvest water from the water-saturated hydrogel.
"Due to the hot climate in Singapore, about 45 million cubic metres of water is lost from reservoirs through evaporation each year. Our hydrogel can convert moisture present in humid air to water without the need for any external energy input. By harnessing the moisture-rich air that is commonly found above water surfaces, we can collect water, which would otherwise be lost to the environment, for different uses," said research team leader Assistant Professor Swee-Ching TAN, who is from the Department of Materials Science and Engineering at the NUS Faculty of Engineering.
The findings of the study have been published online in the scientific journal Advanced Materials earlier this year.
Recovering 'lost' water from water surfaces
Conventional drying agents like silica gel and calcium chloride can only absorb small amounts of water. Extracting water from these agents would require energy intensive processes and high temperatures, and hence this approach is not cost-effective.
The novel hydrogel developed by the NUS team efficiently absorbs water more than four times its original weight. When used repeatedly over multiple absorption-desorption cycles within a day, the amount of water it collects can reach up to 14 times per kilogramme of hydrogel used.
As an added advantage, the novel hydrogel releases water at a relatively lower temperature of between 45 and 50 degrees Celsius, which can be achieved easily with the team's setup. Hence, water could be extracted from the hydrogel using natural sunlight and does not require an external energy source.
Furthermore, while most commercially available drying agents are only suitable for single use, the NUS-developed hydrogel maintains its absorption capability even after 1,000 absorption-desorption cycles.
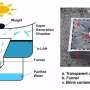
Floating water harvester
As a proof of concept, the research team first built floating devices each containing a small amount of hydrogel placed on a glass plate. These devices were left to float on the water at a beach. Periodically, the water saturated hydrogels were retrieved and transferred to an enclosed glass box which was then exposed to natural sunlight. When exposed to heat, the temperature in the glass box would increase further, allowing moisture within the hydrogels to be released as vapours and later condensed on the walls of the glass box. Condensed water droplets in the glass box were then collected.
The research team further examined the quality of the water harvested from the hydrogels and found that the salt content is almost negligible.
"Unlike processes like desalination which are highly energy intensive, water collected using the novel hydrogel requires less treatment for different uses. Our invention also has potential to be scaled up considerably to function as a floating water-capturing farm. This approach could benefit rural communities where access to clean water remains a challenge. By creatively applying novel solutions, we hope to contribute towards mitigating the global water crisis," said Asst Prof Tan.
Next steps
Taking their research forward, the NUS team is currently looking at ways to optimise the performance of the hydrogel and the water harvesting process. They are also exploring innovative methods to increase absorption capacity of the hydrogel. In addition, the NUS researchers are keen to work with commercial partners to bring this idea to market.
More information: Dilip Krishna Nandakumar et al. Solar Energy Triggered Clean Water Harvesting from Humid Air Existing above Sea Surface Enabled by a Hydrogel with Ultrahigh Hygroscopicity, Advanced Materials (2019). DOI: 10.1002/adma.201806730
Journal information: Advanced Materials
Provided by National University of Singapore