Hydrogels change water and solute dynamics
Hydrogels form tiny, water-filled pores with diameters so small that molecular-level studies can be difficult. Moreover, the motion of water molecules and dissolved ions is so fast that they are difficult to follow. Therefore, little is known about the dynamics of molecular motion within hydrogel structures. Researchers synthesized gel channels and resolved the motion in those channels. They found that molecular dynamics were much slower in hydrogel pores than what is commonly observed in bulk solutions. Within pores, the molecular motion of water was uniform no matter the location within the pore. However, the motion of dissolved ions was much slower close to the polymer fibers that form the pore structures.
Hydrogels have many potential practical uses. These uses range from biomedical applications such as tissue engineering, wound dressings, and contact lenses, to materials for separation membranes in super adsorbants and energy storage devices. Unfortunately, little is known about the gels. This research offers insights about water and dissolved ion motion that could one day lead to better hydrogel designs.
With potential uses ranging from wound dressings to energy storage devices, hydrogels are a promising material. Hydrogels consist of trapped water and ions within a 3-D network of pores. In terms of overall size, hydrogels are particularly compact because much of their structure consists of the water molecules located inside of them. These gels are straightforward to manufacture, and in nature, biological hydrogel structures can form inside and outside of cells. However, scientists do not have a detailed view of the motion of water and dissolved ions inside hydrogel pores at the molecular level.
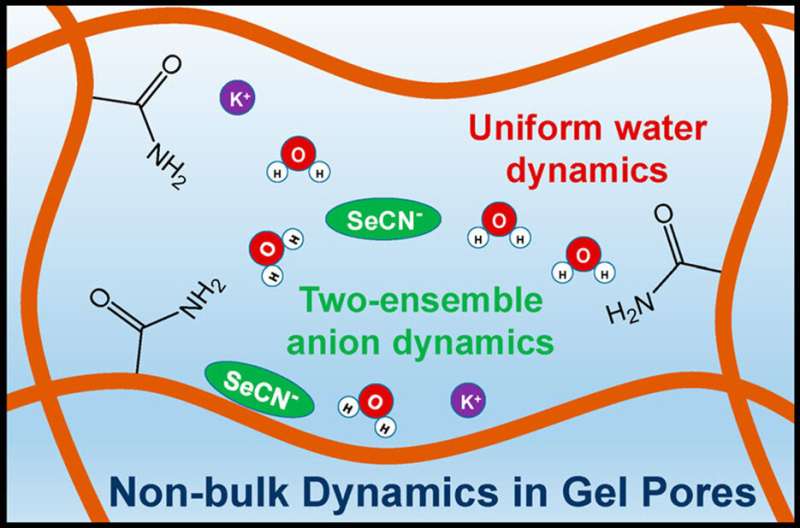
Now, a team of researchers took on these gels. They found that water and solutes (specifically, selenocyanate (SeCN‒)) behave differently inside the gelatinous pores than they would in bulk water. That is, the pores change the dynamics and interactions of water and solute. For example, they found that the network formed by a group of water molecules reorganizes at a slower rate when located inside the pores.
They also found that the dynamics of the water network is the same at any location within the pore. For dissolved ions, this isn't the case because the corresponding dynamics slow closer to the pore walls. These dynamics are challenging to study in hydrogels because the pores are so small and the motions are so fast. This research offers insights about how water and ions move in the gel's pores. One day, this information could lead to better hydrogel designs.
More information: Chang Yan et al. Water Dynamics in Polyacrylamide Hydrogels, Journal of the American Chemical Society (2018). DOI: 10.1021/jacs.8b03547
Journal information: Journal of the American Chemical Society
Provided by US Department of Energy