Direct methane conversion under mild conditions by thermo-, electro- or photocatalysis reviewed
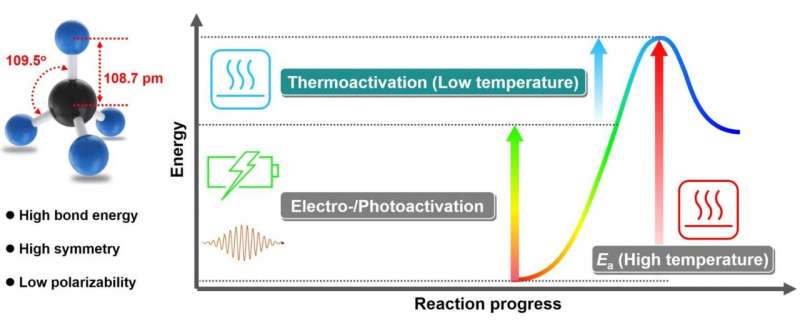
Direct conversion of Earth-abundant methane into value-added chemicals under mild conditions is an attractive technology in response to the increasing industrial demand for feedstocks and the worldwide appeal of energy conservation. Exploring advanced low-temperature C-H activation catalysts and reaction systems is the key to converting methane in a direct and mild manner.
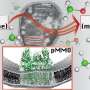
Recently, a research group led by Prof. DENG Dehui from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences reviewed the latest progress in low-temperature methane conversion in thermocatalytic, electrocatalytic, and photocatalytic systems. The study was published in Chem.
"We summarized the typical catalysts employed in various reaction systems, especially the heterogeneous catalysts with noteworthy C-H activation performance," said Prof. DENG.
"The viewpoints on the catalyst design, theoretical simulations, choice of reaction conditions, and method of reaction product analysis were introduced to encourage more viable technology for low-temperature methane conversion in the future," said Prof. DENG.
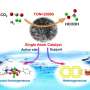
The researchers also pointed out the importance of coupling multiple driving forces from thermal, electrical and solar energy to jointly activate methane by integrating the advantages of these activation pathways in one reaction system.
Prof. DENG's group has been focusing on the development of 2-D material-based catalysts and their applications in the catalytic conversion of energy-related molecules (Nature Nanotechnology, 2016, 11, 218-230; Chemical Reviews, 2019, 119, 1806-1854).
As early as 2015, Prof. DENG and Prof. BAO Xinghe, et al. reported the capability of graphene-confined single iron sites for the catalytic oxidation of complicated hydrocarbons at room temperature (Science Advances, 2015, 1, e1500462).
Notable recent progress by the group includes the finding that graphene-confined single iron atoms could even catalyze methane conversion at room temperature (Chem, 2018, 4, 1902-1910).
These results demonstrate bright prospects for 2-D-based catalysts in the application of C-H activation and other useful catalytic processes.
Journal information: Chemical Reviews , Nature Nanotechnology , Chem , Science Advances
Provided by Chinese Academy of Sciences