Chemists synthesize electrodes for accumulators from coffee grounds
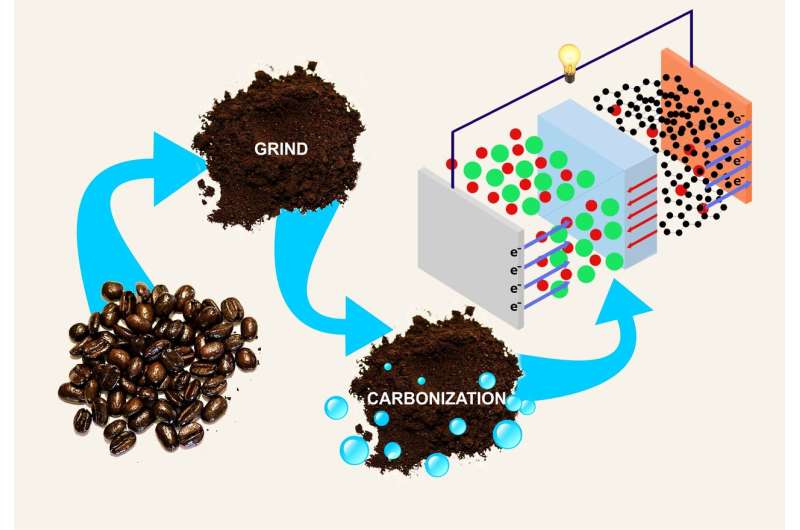
A chemist from RUDN suggested a cheap and eco-friendly way of manufacturing electrodes for lithium-ion batteries from coffee production waste. An article describing this study was published in the Journal of Cleaner Production.
Anodes (negative electrodes) in modern lithium-ion batteries used in electrical appliances and electric cars are most often made of graphite. Research groups are looking for ways to replace it with other carbon materials such as organic waste. Among the candidates were cherry and olive stones, pomace, seaweed and rice husk. In their new work, chemists suggested turning coffee grounds into the necessary carbon material. Around 90 percent of coffee production in the world produces coffee grounds, but this type of waste hasn't been exploited by industry.
The researchers tested a simple method of turning coffee grounds into a useful material without additional chemical processing. The grounds taken from a university canteen were ground once again to make it homogenous, and then baked at 800 degrees C. Electrodes for the electrochemical experiment (13 mm disks) were made from the obtained material and plastic binders mixed at the ratio of 80:20. When tested in an accumulator, they showed the heat storage capacity of 285 mA-h at the current strength of 0.1A. This is a modest value compared to modern accumulators, but a quite high in comparison with anodes made from other types of organic waste. Moreover, their coulombic efficiency (the ratio of the energy given out by an accumulator to that received in the course of charging) is close to 100 percent.
Although the process of manufacturing anodes from coffee grounds turned out to be quite energy-intensive, in the future, it may be more prospective for the use in accumulators than electrodes from cheaper carbon materials. The properties of electrodes made of coffee grounds make them more long-lasting and therefore energy saving.
"Lithium-ion accumulators with our electrodes kept their initial capacity after 100 cycles, losing only 0.23 percent per cycle," says Raphael Luke, director of the Center for Molecular Design and Synthesis of Innovative Compounds for Medicine, and a visiting scholar at RUDN. "Increasing energy storage requirements make our work particularly interesting. It's advantages lie in its simplicity and expediency. This method may be scaled out to industrial levels, as coffee grounds are available all over the world in huge quantities compared to other substances used in the production of lithium-ion batteries."
More information: Fernando Luna-Lama et al. Non-porous carbonaceous materials derived from coffee waste grounds as highly sustainable anodes for lithium-ion batteries, Journal of Cleaner Production (2018). DOI: 10.1016/j.jclepro.2018.10.024
Journal information: Journal of Cleaner Production
Provided by RUDN University