Ceramic holds promise for greener optical devices

A lead-free ceramic that could be used in applications ranging from optical sensors and switches to creams for protecting against ultraviolet (UV) light has been developed by A*STAR researchers.
Ceramics made from potassium sodium niobate (KNN) are promising alternatives to lead-based ceramics in electro-optical applications. However, it is both challenging and costly to improve KNN's performance by ensuring it has a high density, fine-grained, chemically uniform microstructure.
Known as PLZT, lanthanum modified lead zirconate titanate is one of the most widely used electro-optic ceramics. Yet there are serious ecological concerns regarding toxicity to the environment and living organisms once devices made with it are discarded; PLZT contains around 60 per cent of lead (by weight). The search is on to find lead-free replacements for PLZT.
Santiranjan Shannigrahi, and his colleagues from A*STAR's Institute of Materials Research and Engineering and Institute of High Performance Computing, have developed a method for making a KNN based ceramic material that has the potential for replacing PLZT.
"Developing a lead-free, stable ceramic for practical applications was our key aim," explains Shannigrahi. "For some time now KNN has shown promise as a potential alternative to PLZT, but KNN-based ceramics suffer from a number of intrinsic issues, such as the low density of large, cube-shaped particles that allow moisture to be absorbed, making them unstable and therefore unsuitable for practical use."
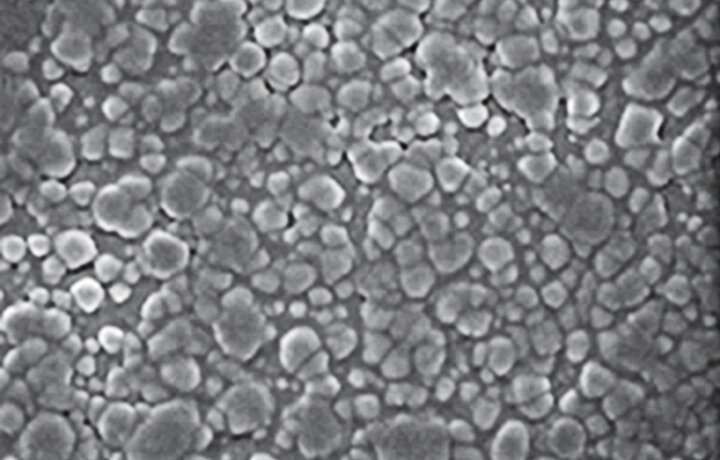
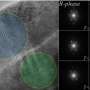
The KNN crystals are modified into nano-sized, nearly spherical particles arranged in a perovskite lattice arrangement. Potassium and sodium ions are located at the corners of the cubed-shaped lattice, oxygen ions at the faces, and niobium ions at the center. The researchers then replaced a proportion of the niobium ions with lanthanum ions, changing the crystal size and structure and creating a completely new material whose magnetic and optical properties can be tuned when exposed to UV.
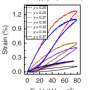
The new material completely absorbs UV light when illuminated, turning into a deep blue color. This is accompanied with a significant increase in magnetization. Interestingly, it returns to its original color and magnetization once illumination ceases.
"These modifications produced a semitransparent ceramic with nano-sized, spherical particles with a density of approximately 98 per cent of the theoretical potential," says Shannigrahi.
The new material could be used in a range of applications, including powerless UV sensors, optical switches and detectors, and for UV protection in sunscreens.
"Our work could lead to a more environmentally friendly alternative to PLZT, and we are now engaging industrial partners for further development," says Shannigrahi.
More information: Santiranjan Shannigrahi et al. Lead-free perovskite ceramics with ultraviolet-tunable optical and magnetic properties at room temperature, Journal of Applied Physics (2018). DOI: 10.1063/1.5034160
Journal information: Journal of Applied Physics