January 9, 2019 feature
Optoacoustic microscopy at multiple discrete frequencies
Optoacoustic imaging powered by short bursts of continuous wave (CW) lasers can stimulate the emission of ultrasound waves inside an animal or in human subjects. The method can noninvasively capture blood flow and produce 3-D images of cellular microarchitecture. Writing in Light: Science & Applications, Stephan Kellnberger and colleagues at the Institute of Biological and Medical Imaging, now report the possibility of obtaining high-fidelity optoacoustic images with cost-effective lasers controlled at multiple frequencies.
The authors experimentally demonstrated the multiple frequency-based, high-fidelity image generation of biological architecture by imaging fish and mouse tissue microvasculature. In the imaging experiments, they superimposed structural details that only appeared at specific frequencies of interest. The authors also non-invasively identified the speed of blood flow in tissue microvasculature by tracking the frequency shifts using the optoacoustic Doppler Effect.
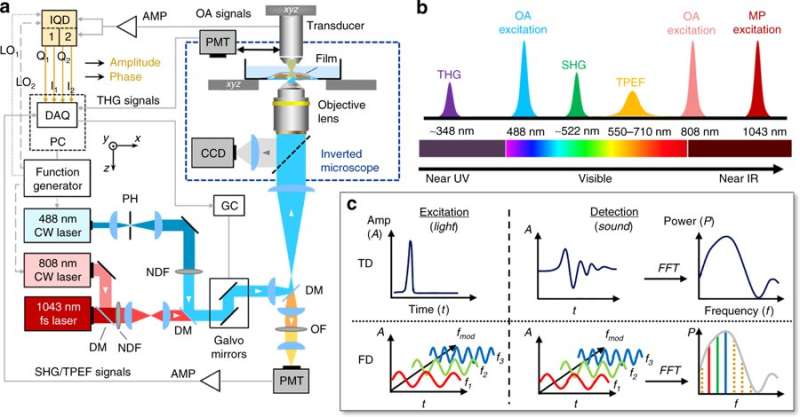
Optoacoustic (photoacoustic) sensing usually requires complex laser technologies. Such techniques can generate nanosecond length (1-100 ns), high-energy short photon pulses that conventionally illuminate transient (short-lived) energy in the time domain (TD). The ultra-short pulses can stimulate the emission of broadband ultrasonic waves, collected in the microsecond range to form optoacoustic images. However, complex laser technology can impose a low-pulse repetition frequency (PRF) and limit the number of wavelengths simultaneously available for spectral imaging. To avoid such limits, Kellnberger et al. developed frequency-domain optoacoustic microscopy (FDOM), in which light intensity can be controlled or modulated at multiple discrete frequencies using cost-effective hardware.
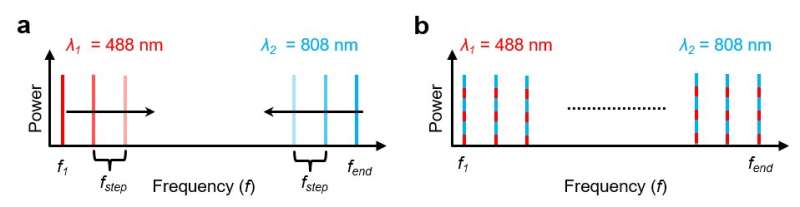
Thus far, optoacoustic imaging has only relied on techniques that detect signals in the time domain (TD) or those that only scan a single frequency at one or two wavelengths in the frequency domain (FD). The present study was a first to conduct in vivo optoacoustic imaging in an animal model via simultaneous illumination with two wavelengths.
The scientists combined FDOM into a hybrid system to examine the relationship between image formation and frequency control. The use of discrete frequencies (a maximum of nine), allowed non-invasive optoacoustic Doppler shift measurements as flow observations in a microfluidic flow chamber in the lab first, and in tissue microvasculature in vivo thereafter. In the study, Kellnberger et al. used two CW diode lasers emitting light at 488 nm and 808 nm for illumination.
The scientists implemented the FDOM, operating in the frequency range of 5-50 MHz, as a hybrid system with multiphoton (MP) microscopy operating at 1043 nm. They then performed two-/three-dimensional imaging based on ultrasound amplitude and phase measurements at multiple frequencies. The amplitude and phase of the generated optoacoustic signals were resolved via demodulation in real time and recorded using an analog-to-digital converter. Due to high repetition rates, the FDOM achieved high signal-to-noise ratios (SNR), leading to the observed high-fidelity images. In total, the study examined the relationship between the modulation frequency, image fidelity and the signal-to-noise-ratio (SNR).
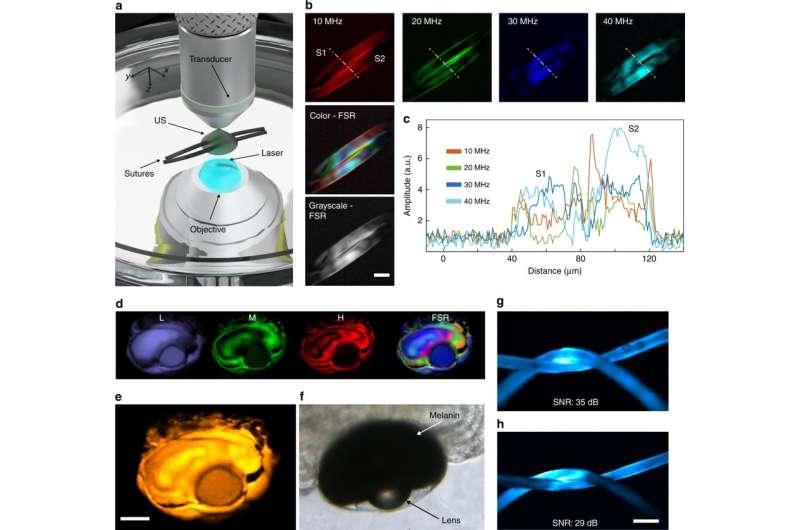
To identify the characteristics of FD photoacoustic imaging, the scientists imaged a pair of crossed sutures in water at two wavelengths (488 nm and 808 nm) and discrete modulation frequencies. The superposition of various frequency contributions carried information of the imaged object (sutures).
To extract information from more complex structures, Kellnberger et al. imaged the eye of 5-day-old wild-type Zebrafish lava ex vivo, using nine modulation frequencies spanning 10-50 MHz in 5-MHz steps. The scientists also compared the SNR (signal-to-noise ratio) between the FDOM method and conventional TD, which varied according to experimental parameters (laser energy, power employed and data acquisition hardware).
Multifrequency amplitude and phase data could thus be processed for 3-D image reconstruction using a Fourier transform based on frequency-space representation (FSR) and time-space representation (TSR). Compared with TSR, the FSR based image reconstruction was computationally faster and did not require data inversion during image reconstruction.
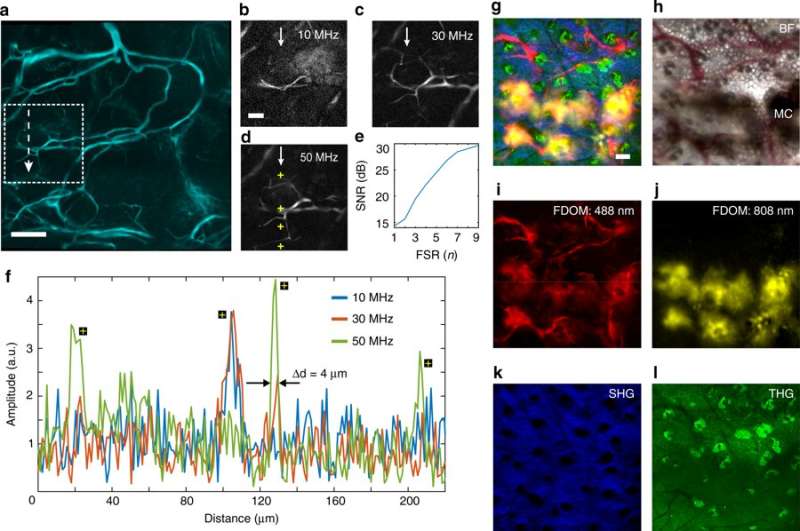
For FDOM-based in vivo tissue imaging, the scientists observed the ear of an anesthetized mouse. They obtained artifact-free images with multiple modulation frequencies that matched spatial frequencies of the imaged object. The scientists used a maximum of nine frequencies in the study. The SNR of the image increased from ~14 dB at a single frequency to ~30 dB at nine frequencies for sharper images.
They then observed a mouse ear containing murine metastatic melanoma cells in vivo as before via synchronized excitation of two wavelengths (488 nm and 808 nm) at separate modulation frequencies. Using combined optoacoustic and optical microscopy, Kellnberger and co-workers were able to efficiently image the tissue features (i.e. vasculature, melanoma cells, collagen and keratinocytes) without conventional fluorescent tags or labels.
Kellnberger et al. then performed FD micro-Doppler (µDoppler) measurements with the setup for the first time in a mouse ear for optoacoustic imaging of microcirculatory blood flow in vivo. Before conducting the intended measurements, the scientists used black carbon particles at varying flow rates of circulation in a microfluidic chip to validate the experimental setup. The µDoppler FDOM was employed to generate a map of microcirculation in a mouse ear thereafter. The microcirculatory blood flow revealed gradually increasing speed from the vessel edge to the core.
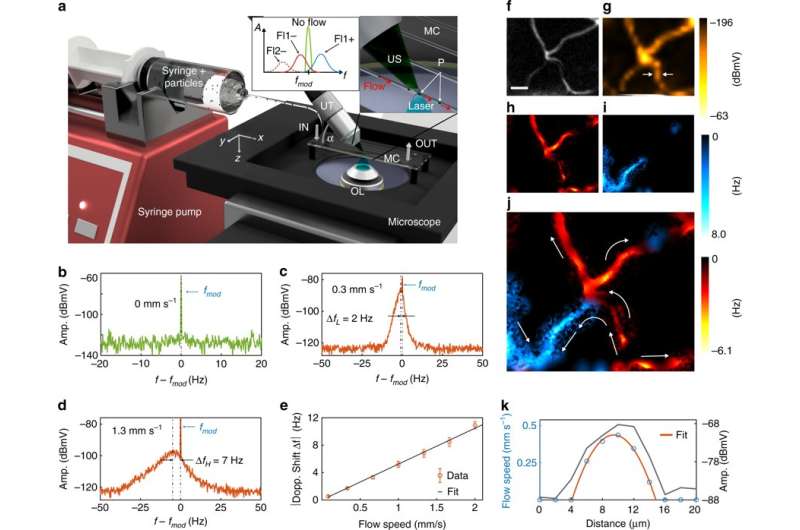
In this way, the study demonstrated the use of frequency-domain optoacoustic microscopy (FDOM) based signal detection and demodulation for the first time. The scientists captured signals of amplitude and phase at multiple frequencies of the imaged object. The collective experimental setup contained inexpensive light sources, simultaneous multiwavelength illumination and direct Doppler-based flow measurements. In future studies, Kellnberger et al. will quantify the modulation frequencies, the imaging depth and increase the image resolution using an improved experimental setup.
More information: Stephan Kellnberger et al. Optoacoustic microscopy at multiple discrete frequencies, Light: Science & Applications (2018). DOI: 10.1038/s41377-018-0101-2
Sergey Telenkov et al. Frequency-domain photothermoacoustics: Alternative imaging modality of biological tissues, Journal of Applied Physics (2009). DOI: 10.1063/1.3116136
George J. Tserevelakis et al. Hybrid multiphoton and optoacoustic microscope, Optics Letters (2014). DOI: 10.1364/OL.39.001819
Vasilis Ntziachristos. Going deeper than microscopy: the optical imaging frontier in biology, Nature Methods (2010). DOI: 10.1038/nmeth.1483
Journal information: Light: Science & Applications , Journal of Applied Physics , Optics Letters , Nature Methods
© 2019 Science X Network