Development of a new fungal production platform C1 for vaccines and therapeutic proteins
VTT and Dyadic International, Inc. today announced that VTT scientists will present the excellent results in their collaboration over the past two years at two international conferences: AAPS PharmSci on November 7th in Washington, D.C., and PEGS Europe on November 14th in Lisbon.
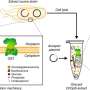
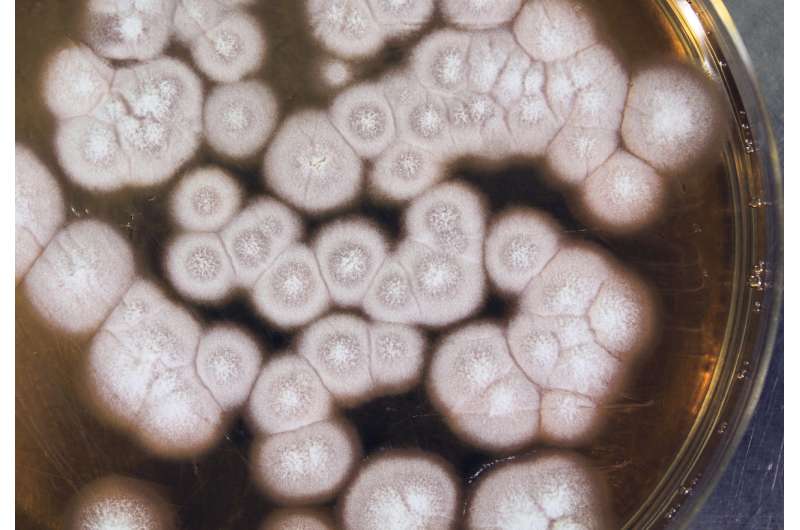
The two-year research and development collaboration has successfully positioned Dyadic's C1 Myceliophthora thermophila fungus as a potential production system for therapeutic proteins, vaccines and high value primary and secondary metabolites. Dyadic's industrially proven filamentous fungal strain of Myceliophthora thermophila, nicknamed C1, is known as an extremely robust and efficient protein production organism in the enzyme industry. In addition to being able to produce high levels of proteins, C1 produces glycan structures more similar to the human glycan forms than most other non-mammalian production hosts, making the glycoengineering work carried out in the project quicker and easier. Dyadic has developed and used its C1 production platform to produce industrial enzymes since the late 1990's.
VTT and Dyadic will present data regarding the development of an extremely low protease and high-producing platform strain that has resulted in the production of stable therapeutic antibodies with a final production rate of over 2.6 grams/litre/day. Furthermore, several difficult-to-express proteins, such as vaccine antigens and bispecific antibodies, have been produced at good levels in C1 as well as a specific primary metabolite through the metabolic engineering of C1. Additionally, specific information will be presented on the glycoengineering work to humanize the glycosylation machinery of C1 that has also generated promising results.
In addition, from the start of 2017 until now, Dyadic and VTT have established research projects with 10 groups including top pharmaceutical companies such as Mitsubishi Tanabe Pharma and Sanofi-Aventis. So far, research milestones have been successfully achieved in all the completed projects.

"We are very excited about the impressive progress that was made by the VTT project team. I am sure that the high skills and experience of the VTT scientists in strain development will continue to help us to reach our ambitious goals of C1 as an important production platform in the pharmaceutical world," said Ronen Tchelet, Ph.D.
The pharma industry is producing therapeutic proteins mainly in the CHO (Chinese hamster ovary) cell system, but the current production processes are quite time consuming and expensive. In the Dyadic-VTT collaboration, rapid progress and significant advances have been made to the industrialized C1 host cell. This C1 gene expression platform provides the possibility to bring affordable medicines to patients sooner and at a lower cost while reducing the financial burden on the global healthcare system.
In the C1 development program, VTT is utilizing its strong competence in fungal molecular biology and bioprocesses that has been accumulated during more than 30 years of research on a variety of fungi. This collaboration has shown that the C1 system has excellent prospects for becoming a game changing platform for therapeutic protein production.
"The VTT project team is committed to making the production of new, better, lower cost medicines in C1 a reality in the future. We have been highly impressed with the great ability of the C1s fungus to produce proteins with high efficiency and good quality," said Dr. Markku Saloheimo, Senior Principal Scientist at VTT.
Provided by VTT Technical Research Centre of Finland