The tightest non-aminoglycoside ligand for the bacterial ribosomal RNA A-site
A research group at Tohoku University has made a significant discovery with positive implications for the development of bacteria-fighting drugs. The aminoacyl-tRNA site (A-site) of the 16S RNA decoding region in the bacterial ribosome looks promising for a new era of antibiotic drug development.
Traditional aminoglycoside antibiotics are problematic given their high toxicity and the potential for resistance development. The research at Tohoku University focused on bacterial A-site binding small ligands whose structures are distinct from the aminoglycoside family, which offer potential for the development of novel drugs that treat bacterial infections with a reduction in the problems associated with traditional antibiotics.
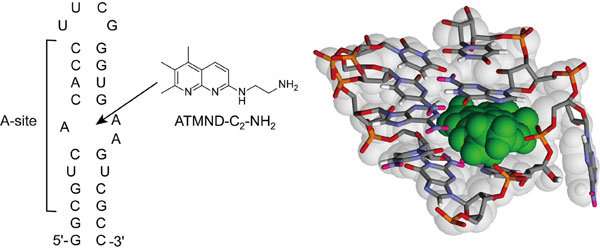
The research group led by Dr. Seiichi Nishizawa and Dr. Yusuke Sato (Department of Chemistry, Graduate School of Science) has reported a novel small ligand, ATMND-C2-NH2 that has the tightest binding affinity for the bacterial A-site among the non-aminoglycoside ligands.
ATMND-C2-NH2 shows a significant fluorescent quenching response upon selective binding to the internal loop of the bacterial (Escherichia coli) A-site-containing model RNA.
ATMND-C2-NH2 has also proven useful as an indicator for assessing ligand/A-site interactions.
The results obtained by the research group offer a rational basis for the generation of novel A-site binding ligands with a view toward novel antibiotics with less toxicity and minimum resistance development.
More information: Yusuke Sato et al. Fluorescent Trimethylated Naphthyridine Derivative with an Aminoalkyl Side Chain as the Tightest Non-aminoglycoside Ligand for the Bacterial A-site RNA, Chemistry - A European Journal (2018). DOI: 10.1002/chem.201802320
Journal information: Chemistry – A European Journal
Provided by Tohoku University