RNA-protein network may explain why melanoma grows more
With five-year survival rates being around 30 percent for patients with distant metastatic disease, cutaneous melanoma is the leading cause of skin cancer-related deaths. The major causes of the low survival rate for melanoma patients are the limited number of options for patients lacking the BRAF mutation and the intrinsic and acquired resistance to existing therapies. It is therefore essential to develop new therapeutic strategies to eradicate resistant cells and/or target patients irrespective of their driver mutations.
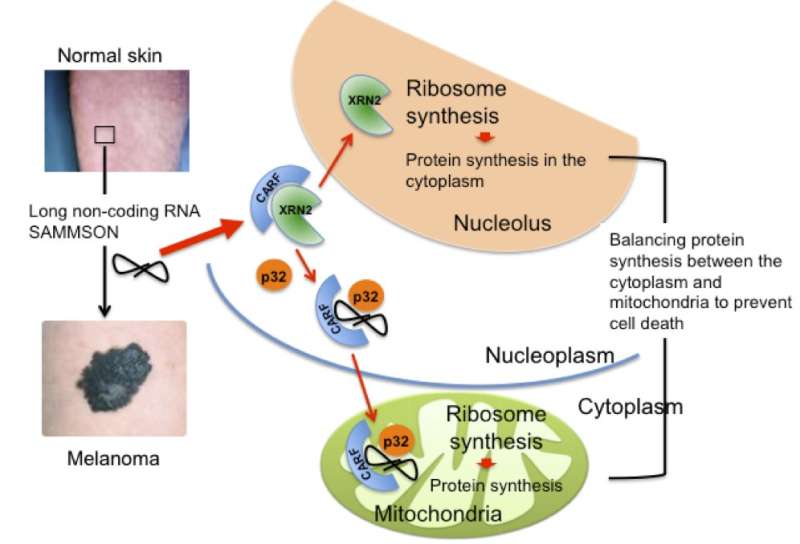
A collaboration led by scientists from KU Leuven, Belgium, with Tokyo University of Agriculture and Technology (TUAT), Japan, revealed a new way to fight melanoma. They report that a melanoma-specific long non-coding RNA, named SAMMSON, interacts with the protein CARF to properly coordinate protein synthesis in both the cytosol and mitochondria of melanoma cells. This mechanism ensures the maintenance of proteostasis during cell growth, thus avoiding the induction of cell death.
"We identified a long non-coding RNA named SAMMSON expressed in the vast majority (>90 percent) of melanoma patients and never detected in normal adult tissues," said co-senior author Dr. Eleonora Leucci, LKI Leuven Cancer Institute, KU Leuven. "In melanoma, SAMMSON is essential for proper localization of the protein p32 into the mitochondria, where it is essential for ribosome biogenesis and protein synthesis."
"We reported that protein CARF presents in the nucleoplasm, indirectly reduces the ribosome synthesis in the nucleolus through directly binding to another protein XRN2," said co-author Dr. Hideaki Ishikawa, Department of Agriculture, TUAT, Tokyo, Japan. "In the nucleolus, XRN2 acts as an enzyme that increases the ribosome synthesis. But its activity and localization are regulated by CARF whose binding keeps XRN2 away from the nucleolus."
These independent observations where connected later by first author MSc Roberto Vendramin who found that SAMMSON binds to CARF. This observation triggered the international collaboration that led to a work published in the journal Nature Structural & Molecular Biology.
"We found that in melanoma, a portion of CARF is sequestered into the cytoplasm where it interacts with p32 in a SAMMSON-dependent manner" said Dr. Eleonora Leucci.
"Released from the CARF binding, XRN2 translocates from the nucleoplasm to the nucleolus where it promotes ribosome biogenesis. Concertedly the complex CARF-SAMMSON-p32, moves to the mitochondria and boosts the mitochondrial ribosome synthesis."
"We concluded that, in melanoma cells, SAMMSON increases the ribosome synthesis concertedly in the nucleolus and mitochondria," said Dr. Leucci. "Cells have a mechanism to sense the abnormality if the ribosome synthesis is altered in either the nucleolus or mitochondria, and induce cell death. However, when this process is altered in both compartments the cell is unable to recognize the insult. Therefore, by hacking concertedly both ribosome biogenesis machinery, SAMMSON makes the cell insensitive to the damage and promotes melanoma cell growth"
Hence, a substance that inhibits SAMMSON or/and CARF is expected to be a new therapeutic reagent for melanoma.
More information: Roberto Vendramin et al, SAMMSON fosters cancer cell fitness by concertedly enhancing mitochondrial and cytosolic translation, Nature Structural & Molecular Biology (2018). DOI: 10.1038/s41594-018-0143-4
Journal information: Nature Structural & Molecular Biology
Provided by Tokyo University of Agriculture and Technology