Breakthrough in blending metals—precise control of multimetallic one-nanometer cluster formation achieved
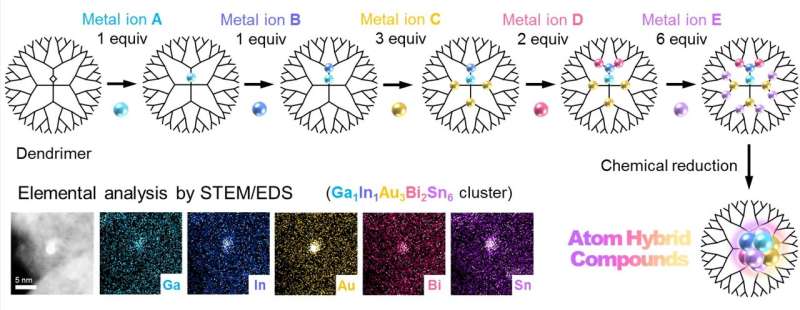
Researchers in Japan have found a way to create innovative materials by blending metals with precision control. Their approach, based on a concept called atom hybridization, opens up an unexplored area of chemistry that could lead to the development of advanced functional materials.
Multimetallic clusters—typically composed of three or more metals—are garnering attention as they exhibit properties that cannot be attained by single-metal materials. If a variety of metal elements are freely blended, it is expected that as-yet-unknown substances are discovered and highly-functional materials are developed. So far, no one had reported the multimetallic clusters blended with more than four metal elements so far because of unfavorable separation of different metals. One idea to overcome this difficulty is miniaturization of cluster sizes to one-nanometer scale, which forces the different metals to be blended in a small space. However, there was no way to realize this idea.
A team, including Takamasa Tsukamoto, Takane Imaoka, Kimihisa Yamamoto and colleagues, has developed an atom hybridization method, which has realized the first-ever synthesis of multimetallic clusters consisting of more than five metal elements with precise control of size and composition. This method employs a dendrimer template that serves as a tiny "scaffold" to enable controlled accumulation of metal salts. After precise uptake of the different metals into the dendrimer, multimetallic clusters are obtained by chemical reduction. In contrast, a conventional method without the dendrimer yields enlargement of cluster sizes and separation of different metals. The team successfully demonstrated the formation of five-element clusters composed of gallium (Ga), indium (In), gold (Au), bismuth (Bi) and tin (Sn), as well as iron (Fe), palladium (Pd), rhodium (Rh), antimony (Sb) and copper (Cu), and a six-element cluster consisting of Ga, In, Au, Bi, Sn and platinum (Pt). Additionally, they hint at the possibility of making clusters composed of eight metals or more.

This atom hybridization method using the dendrimer template can synthesize ultrasmall multimetallic clusters with precise control of size and composition. There are more than 90 metals in the periodic table. With infinite combinations of metal elements, atomicity and composition, this method will open up a new field in chemistry on a one-nanometer scale. The current study marks a major step forward in creating such as-yet-unknown innovative materials.
More information: Takamasa Tsukamoto et al, Atom-hybridization for synthesis of polymetallic clusters, Nature Communications (2018). DOI: 10.1038/s41467-018-06422-8
Journal information: Nature Communications
Provided by Tokyo Institute of Technology



















