A new method of transporting ions through cell membranes based on a single amino acid
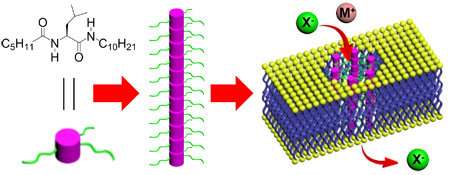
Researchers from A*STAR's Institute of Bioengineering and Nanotechnology (IBN) have successfully developed a unique family of pore-forming monopeptides based on a single amino acid. Other pore-forming peptides typically consist of up to fifty amino acids. The research team, led by IBN Team Leader and Principal Research Scientist Dr Huaqiang Zeng, has created a new paradigm for the efficient transportation of important ions across cellular membranes. This has the potential to tackle diseases where this kind of ion transport has malfunctioned, such as cystic fibrosis.
Pore-forming peptides form channels through cellular membranes for the transportation of ions such as chlorides and iodides in and out of a cell. The formation of these channels are crucial for the precise regulation of our bodies' physiological processes, cellular defenses and immune responses. Patients with cystic fibrosis suffer from a defective gene that reduces the transport of such ions, thus dehydrating the mucus layer in the lungs. This results in a build-up of thick and viscous mucus that causes breathing difficulties.
The IBN researchers focused on studying artificial pore-forming monopeptides based on a single amino acid. These could form channels of pore sizes smaller than 1.0 nm in diameter, while remaining capable of transporting negatively-charged ions, including chloride, efficiently across the membrane. Designing artificial ion channels is an area of increasing interest, particularly channels that can self-assemble in a controlled manner from simple molecular precursors. Many current systems utilise complex, high molecular weight components, which greatly limit their potential for future therapeutic applications. This unique breakthrough is one of the most exciting advances in the field of artificial ion channels in recent years. It presents opportunities to design novel small molecule-based artificial ion channels which can result in better therapies for ion channel dysfunctions, and anti-bacterial or anti-cancer agents.
Dr Ichiro Hirao, Covering Executive Director of IBN, said, "The IBN research team originally studied ion-transporting molecules designed to function as gelators to coalesce oil spills in water. Through this, they discovered that one of the gelators was of significant toxicity, causing them to explore whether the molecule might self-assemble into a pore-like structure in the cell membrane and cause an imbalance of ions. This hypothesis turned out to be true and led to a completely new principle of the molecular design of ion channels."
The research results were published in the Journal of the American Chemical Society on 21 June 2018.
More information: Changliang Ren et al. Pore-Forming Monopeptides as Exceptionally Active Anion Channels, Journal of the American Chemical Society (2018). DOI: 10.1021/jacs.8b04657
Journal information: Journal of the American Chemical Society





















